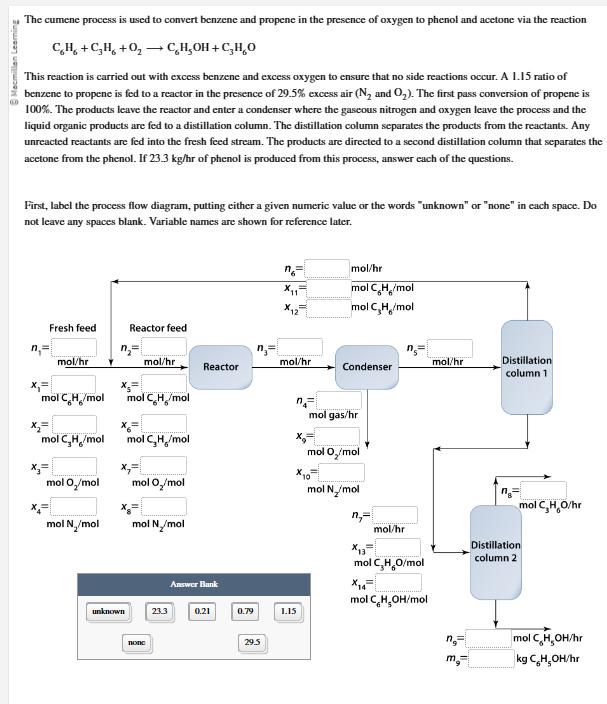
Macmillan Learning The cumene process is used to convert benzene and propene in the presence of...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Macmillan Learning The cumene process is used to convert benzene and propene in the presence of oxygen to phenol and acetone via the reaction CH+CH+0,CH,OH+CHO This reaction is carried out with excess benzene and excess oxygen to ensure that no side reactions occur. A 1.15 ratio of benzene to propene is fed to a reactor in the presence of 29.5% excess air (N2 and O2). The first pass conversion of propene is 100%. The products leave the reactor and enter a condenser where the gaseous nitrogen and oxygen leave the process and the liquid organic products are fed to a distillation column. The distillation column separates the products from the reactants. Any unreacted reactants are fed into the fresh feed stream. The products are directed to a second distillation column that separates the acetone from the phenol. If 23.3 kg/hr of phenol is produced from this process, answer each of the questions. First, label the process flow diagram, putting either a given numeric value or the words "unknown" or "none" in each space. Do not leave any spaces blank. Variable names are shown for reference later. n mol/hr mol C H/mol mol CH/mol Fresh feed Reactor feed n = n = n= mol/hr mol/hr mol/hr mol/hr Distillation Reactor Condenser column 1 x = x= mol C.H/mol mol C. Ho/mol mol gas/hr mol C,H/mol mol CH/mol mol O/mol mol O/mol mol O/mol mol N/mol mol C,H,O/hr mol N/mol mol N/mol Answer Bank unknown 23.3 0.21 0.79 1.15 none 29.5 mol/hr mol C,HO/mol mol CHOH/mol Distillation column 2 n mol C HOH/hr kg CHOH/hr What is 1, the molar flow rate of fresh feed into the process? What are the mole fractions of CH (x1), CH, (x2), O (x3), and N2 (x4) in the fresh feed stream? x= x2= x3= x4= What is no n = the molar flow rate of the recycle stream? mol/hr mol CH/mol mol CH/mol mol O/mol mol N/mol mol/hr Macmillan Learning The cumene process is used to convert benzene and propene in the presence of oxygen to phenol and acetone via the reaction CH+CH+0,CH,OH+CHO This reaction is carried out with excess benzene and excess oxygen to ensure that no side reactions occur. A 1.15 ratio of benzene to propene is fed to a reactor in the presence of 29.5% excess air (N2 and O2). The first pass conversion of propene is 100%. The products leave the reactor and enter a condenser where the gaseous nitrogen and oxygen leave the process and the liquid organic products are fed to a distillation column. The distillation column separates the products from the reactants. Any unreacted reactants are fed into the fresh feed stream. The products are directed to a second distillation column that separates the acetone from the phenol. If 23.3 kg/hr of phenol is produced from this process, answer each of the questions. First, label the process flow diagram, putting either a given numeric value or the words "unknown" or "none" in each space. Do not leave any spaces blank. Variable names are shown for reference later. n mol/hr mol C H/mol mol CH/mol Fresh feed Reactor feed n = n = n= mol/hr mol/hr mol/hr mol/hr Distillation Reactor Condenser column 1 x = x= mol C.H/mol mol C. Ho/mol mol gas/hr mol C,H/mol mol CH/mol mol O/mol mol O/mol mol O/mol mol N/mol mol C,H,O/hr mol N/mol mol N/mol Answer Bank unknown 23.3 0.21 0.79 1.15 none 29.5 mol/hr mol C,HO/mol mol CHOH/mol Distillation column 2 n mol C HOH/hr kg CHOH/hr What is 1, the molar flow rate of fresh feed into the process? What are the mole fractions of CH (x1), CH, (x2), O (x3), and N2 (x4) in the fresh feed stream? x= x2= x3= x4= What is no n = the molar flow rate of the recycle stream? mol/hr mol CH/mol mol CH/mol mol O/mol mol N/mol mol/hr
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Formaldehyde is produced from methanol in a catalytic oxidation reactor. The following reactions take place: CH3OH HCHO + H2 2H2 + O2 2H2O A stream of methanol is joined by a recycle stream, also...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Given the functions f(x) = 2x and g(x) = x 1) Find the points of intersection of the curves and plot the graphs of the functions. 2) Determine the area bounded by the curves in the interval [-1,3] 3)...
-
Let A be a square matrix and P a permutation matrix of the same size. (a) Explain why the product APT has the effect of applying the permutation defined by P to the columns of A. (b) Explain the...
-
From the following, calculate (a) Net sales. (b) Gross profit. (c) Net income. Data Sales, $22,000; Sales Discounts, $500; Sales Returns and Allowances, $250; Cost of Goods Sold, $13,200; Operating...
-
Zappos.com is a popular website known mainly for its discounted shoe sales. In 2012, a hacker hacked into the Zappos website in an effort to obtain the personal account information of Zappos...
-
1. Toula and Ian Miller of Lincoln, Nebraska, recently suffered a fire in their home. The fire, which began in a crawl space at the back of the house, caused $24,000 of damage to the dwelling. The...
-
The figure shows a schematic diagram of a simple mass spectrometer, consisting of a velocity selector and a particle detector and being used to separate singly ionized atoms (q+e 1.602e-19 C) of gold...
-
Q: The rigid bar CDE is attached to a pin support at E and rests on the 30 mm diameter brass cylinder BD. A 22-mm-diameter steel cylinder AC passes through a hole in the bar and is secured by a nut...
-
What 10 elements are included in the study of AIS?
-
Where are error and exception routines shown on systems flowcharts?
-
Why is the year-end adjusting entry needed for workers compensation insurance?
-
Are the terms system and subsystem synonymous? Explain your answer.
-
Describe the stages of an automated accounting process.
-
Select a company Sears Holdings Corporation and obtain its financial reports. Write a paper on risk factors as identified for all of the major business transactions conducted by the company. Analyze...
-
Information graphics, also called infographics, are wildly popular, especially in online environments. Why do you think infographics continue to receive so much attention? How could infographics be...
-
A project developed in PL/1 is expected to take 30 months. Assuming the same ratios as those shown in the table below, compare the duration, level of effort, and software size between a project...
-
What is the difference between tabular output and zoned output?
-
Develop a questionnaire for mass employee distribution based on your findings from the previous interviews. Why are we completing the analysis with an anonymous survey?

Study smarter with the SolutionInn App


