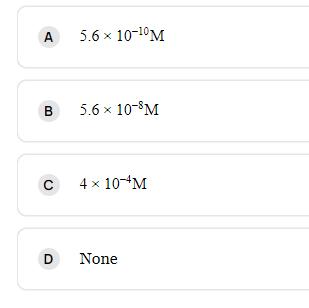
Molar solubility of Ca(OH)2 in a solution that has a pH of 12.[KSP[Ca(OH)2]=5.61012] A B C 5.6
Fantastic news! We've Found the answer you've been seeking!
Question:
Molar solubility of Ca(OH)2 in a solution that has a pH of 12.[KSP[Ca(OH)2]=5.6×10−12]
Related Book For 

Fundamentals of Analytical Chemistry
ISBN: 978-0495558286
9th edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Posted Date: