Question: Molecular Formula: C 3 H 6 O 2 Mass SpectrumData: UV-VIS Data: Molecular Ion (M + ) 74 m/z No significant UV absorption > 220
Molecular Formula: C3H6O2
Mass SpectrumData: UV-VIS Data:
Molecular Ion (M+) 74m/z No significant UV absorption > 220 nm.
Fragment 43 m/z (base peak)
Answer the following questions.
11. What is the degree of unsaturation for this compound?
a. 0
b. 1
c. 2
d. 3
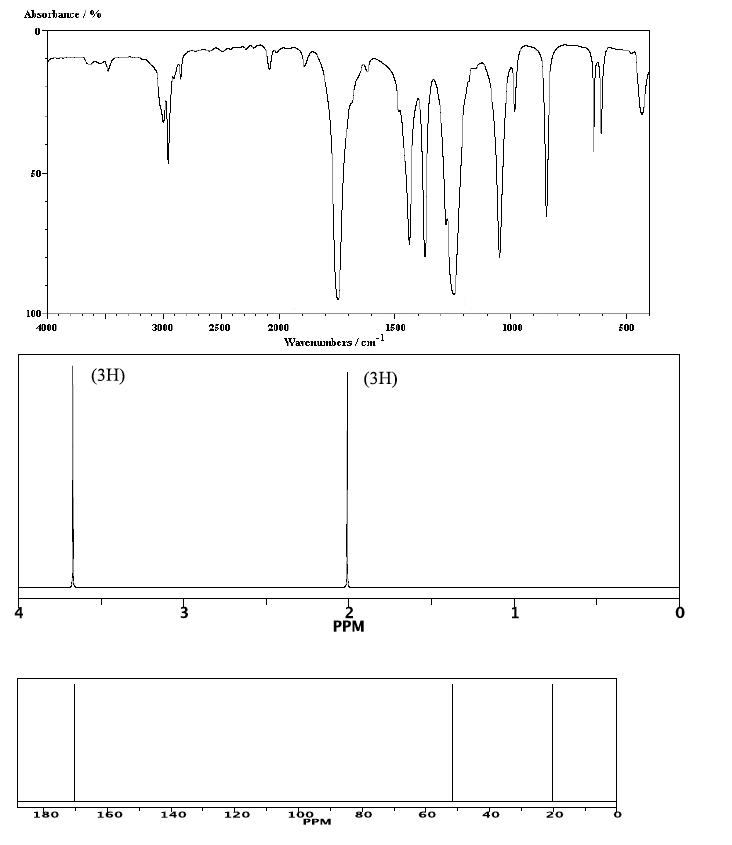
12. The peak at 1746 cm-1 on the IR spectrumtells us the compound contains a(n)
a. hydroxyl group
b. primary amine
c. carbonyl group
d. aromatic ring
13. The sharp intense peak at 1246 cm-1 is usuallyindicative of _____________ stretch.
a. carbon-carbon single bond
b. carbon-nitrogen single bond
c. carbon-hydrogen single bond
d. carbon-oxygen single bond
14. Based on the information from the IR spectrum, themajor functional group in this compound is a(n) _____________.
a. alcohol
b. carboxylic acid
c. ester
d. aldehyde
15. Based on the 1H NMR how many sets ofnon-equivalent protons are in this molecule?
a. 1
b. 2
c. 3
d. 4
16. The most deshielded peak in the 13C NMR ischaracteristic of the ______________ group.
a. vinyl carbon
b. sp-hybridized carbon atoms
c. carbonyl carbon atom
d. aromatic carbon atoms
17. The base peak at m/z 43 in the mass spectrum is dueto the loss of ___________ group.
a. OCH3
b. C=O group
c. CH3 group
d. O=CCH3
18. The peak at 3.4 ppm in the 1H NMR is moredownfield than the peak at 2.0 ppm because it is attached to_______________.
a. another carbon atom
b. an oxygen atom
c. more hydrogen atoms
d. more carbon atoms
19. The molecular ion peak at m/z 74 tells us the______________ of the compound.
a. molecular formula
b. melting point
c. molecular weight
d. empirical formula
20. The name and structure of the compound that best fitsthis data is
Absorbance / % 0 50- 100- 4000 180 (3H) 160 3000 -M 140 2500 120 2000 nam Warenumbers/cm 100 PPM 1500 (3H) 2 PPM 18 1000 T 500
Step by Step Solution
3.43 Rating (169 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


