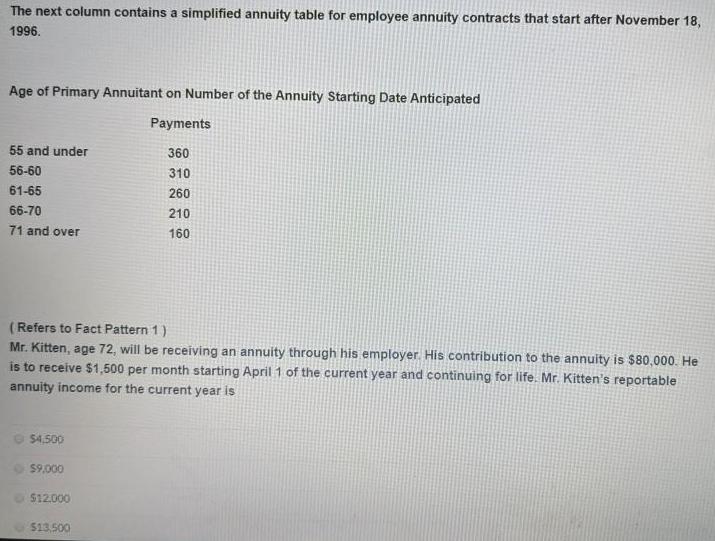
The next column contains a simplified annuity table for employee annuity contracts that start after November...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The next column contains a simplified annuity table for employee annuity contracts that start after November 18, 1996. Age of Primary Annuitant on Number of the Annuity Starting Date Anticipated Payments 55 and under 360 56-60 310 61-65 260 66-70 210 71 and over 160 (Refers to Fact Pattern 1) Mr. Kitten, age 72, will be receiving an annuity through his employer. His contribution to the annuity is $80,000. He is to receive $1,500 per month starting April 1 of the current year and continuing for life. Mr. Kitten's reportable annuity income for the current year is $4.500 $9,000 $12.000 $13.500 The next column contains a simplified annuity table for employee annuity contracts that start after November 18, 1996. Age of Primary Annuitant on Number of the Annuity Starting Date Anticipated Payments 55 and under 360 56-60 310 61-65 260 66-70 210 71 and over 160 (Refers to Fact Pattern 1) Mr. Kitten, age 72, will be receiving an annuity through his employer. His contribution to the annuity is $80,000. He is to receive $1,500 per month starting April 1 of the current year and continuing for life. Mr. Kitten's reportable annuity income for the current year is $4.500 $9,000 $12.000 $13.500
Expert Answer:
Answer rating: 100% (QA)
Solution Answer 9000 Total Contribution to annuity 80000 Total anticpa... View the full answer

Related Book For 

Corporate Financial Accounting
ISBN: 978-1133952411
12th edition
Authors: Carl S. Warren, James M. Reeve, Jonathan E. Duchac
Posted Date:
Students also viewed these accounting questions
-
MZE Manufacturing Company has a normal plant capacity of 37,500 units per month. Because of an extra-large quantity of inventory on hand, it expects to produce only 30,000 units in May. Monthly fixed...
-
Mr Smith owns a small start-up where he will be producing in-house made pharmaceutical products. Mr Smith recently moved from Edmonton, AB to Surrey, BC. He will be setting up his pharmaceutical...
-
Vernon Company is considering the replacement of some of its manufacturing equipment. Information regarding the existing equipment and the potential replacement equipment follows. Existing Equipment...
-
Let z(k) denote the k-year continuously compounded zero-coupon yield for the current term structure. You are given that z(1) = 0.035, z(2) = 0.041, z(3) = 0.045, z(4) = 0.049, z(5) = 0.051, z(6) =...
-
On February 1, 2007, Selma purchased a $50,000 compound-interest CSB. The interest rate on the CSB was 1.55% for each of the first two years and 2.675% for the third year. What was the total interest...
-
Trieste Toy Company manufactures only one product, Robot Ranger. The company uses a standard cost system and has established the following standards per unit of Robot Ranger: During November, the...
-
Refer to the information in Exercise 22-12. Assume that each of the companys divisions has a required rate of return of 7%. Compute residual income for each division. Data From Exercise 22-12 A food...
-
Bullseye Department store is a discount retailer of general merchandise in the Southeastern United States. The company owns more than 50 stores in Florida, Georgia, South Carolina, and Tennessee that...
-
Exercise 1 (8 points). Prove that, for all n = N, (Hint:) you may try by induction. 5|(n-n). Exercise 2 (9 points). Solve the following systems of congruences. (a) (b) x = 6 (mod 10) x = 11 (mod 15)...
-
Get It Right, CPAs, has been retained to review its client's corporate formation calculations for 20XX. Maria, Roger, and Novak created Grassroots Tennis, Inc. (GTI), which began operations on March...
-
Wren Pork Company uses the value basis of allocating joint costs in its production of pork products. Relevant information for the current period follows: Product Pounds Price/lb. 3,360 $5.00 Loin...
-
5. A man pulls a crate of mass 10 kg with a constant speed. The crate is on frictionless bearings up a ramp that is h = 5 m high and angled at 30. (Hint: Remember the direction of force and...
-
A 12 g steel ball bearing rolls to the right at 68 cm/s and makes an elastic head-on collision with another steel ball bearing with a mass of 11 g that is initially at rest. After the collision, the...
-
6. A 16.00 m Three roller-coaster carts with a combined mass of 1250 kg have a speed of 15.0 m/s nearing position D. Ground B 8.50 m C D 2.50 m ODLB As the carts approach position D they are stopped...
-
Show that the total mechanical energy of a satellite (mass m) orbiting at a distance r from the center of the Earth (mass ME) is E = 1 GMME 2 r if U = 0 at r = o. (b) Show that although friction...
-
2. The mechanism shown in the figure consists of a particle m which is connected to a hinge A by an ideal rod with length . Particle m slides in a frictionless vertical slot of a block with mass M...
-
Sam and Ronnie each invest $500,000 per t-0 when the risk-free rate (for all maturities) is 5%: Sam has purchased $500,000 of 6-year zero coupon bonds, where each bond has a face value of...
-
Construct a 4 x 25 design confounded in two blocks of 16 observations each. Outline the analysis of variance for this design.
-
The following accounts, with the balances indicated, appear in the ledger of Garcon Co. on December 1 of the current year: The following transactions relating to payroll, payroll deductions, and...
-
The amounts of the assets and liabilities of Wilderness Travel Service at April 30, 2014, the end of the current year, and its revenue and expenses for the year are listed below. The retained...
-
Complete the following table by indicating for (a) through (g) whether the proper answer is debit or credit: Normal Decrease Balance Account Purchases Purchases Discounts Purchases Returns and...
-
An atom loses an electron to another atom. Is this an example of a physical or chemical change? (a) chemical change involving the formation of ions (b) physical change involving the formation of ions...
-
Aluminum ions carry a 3+ charge, and chloride ions carry a 1- charge. What is the chemical formula for the ionic compound aluminum chloride? (a) Al 3 Cl (b) AlCl 3 (c) Al 3 Cl 3 (d) AlCl
-
Why are ores so valuable? (a) They are sources of naturally occurring gold. (b) Metals can be efficiently extracted from them. (c) They tend to occur in scenic mountainous regions. (d) They hold many...

Study smarter with the SolutionInn App


