4) Use Table 2.3 from your text book to determine the physical state (solid, liquid, or...
Fantastic news! We've Found the answer you've been seeking!
Question:

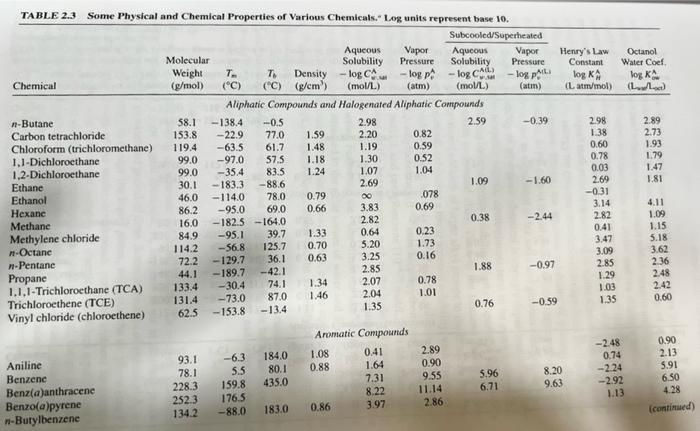
Transcribed Image Text:
4) Use Table 2.3 from your text book to determine the physical state (solid, liquid, or gas) of the following chemicals: Ethane, Ethanol, Hexane, Methane, Methanol, and Naphthalene. Note: Just do not state your answer, show how you determined the physical state of each of these pollutants using relevant physical properties listed in Table 2.3. TABLE 2.3 Some Physical and Chemical Properties of Various Chemicals. Log units represent base 10. Subcooled/Superheated Chemical n-Butane Carbon tetrachloride Chloroform (trichloromethane) 1,1-Dichloroethane 1,2-Dichloroethane Ethane Ethanol Hexane Methane Methylene chloride n-Octane n-Pentane Propane 1.1.1-Trichloroethane (TCA) Trichloroethene (TCE) Vinyl chloride (chloroethene) Aniline Benzene Benz(a)anthracene Benzo(a)pyrene n-Butylbenzene Molecular Weight T To Density (g/mol) (°C) (°C) (g/cm³) 58.1 153.8 119.4 99.0 99.0 Aqueous Vapor Solubility Pressure -log CA -log p (atm) F (mol/L) Aliphatic Compounds and Halogenated Aliphatic Compounds 2.98 2.59 2.20 1.19 1.30 1.07 2.69 30.1 46.0 86.2 16.0 84.9 114.2 72.2 -138.4 -0.5 -22.9 77.0 61.7 -63.5 -97.0 -35.4 -183.3 -114.0 -95.0 -182.5 -164.0 -95.1 -56.8 -129.7 44.1 -189.7 -42.1 133.4 -30.4. 131.4 62.5 57.5 83.5 -88.6 78.0 0.79 69.0 0.66 93.1 -6.3 78.1 5.5 228.3 159.8 252.3 176.5 134.2 -88.0 39.7 125.7 36.1 74.1 -73.0 87.0 -153.8 -13.4 184.0 80.1 435.0 1.59 1.48 1.18 1.24 183.0 1.33 0.70 0.63 1.34 1.46 1.08 0.88 ∞0 3.83 2.82 0.64 5.20 3.25 2.85 Aromatic Compounds 0.41 1.64 7.31 8.22 3.97 0.86 2.07 2.04 1.35 0.82 0.59 0.52 1.04 078 0.69 0.23 1.73 0.16 0.78 1.01 2.89 0.90 9.55 11.14 2.86 Aqueous Vapor Solubility Pressure -log CA -log p (mol/L) 1.09 0.38 1.88 0.76 5.96 6.71 (atm) -0.39 -1.60 -2.44 -0.97 -0.59 Henry's Law Constant log KA (L atm/mol) 8.20 9.63 2.98 1.38 0.60 0.78 0.03 2.69 -0.31 3.14 2.82 0.41 3.47 3.09 2.85 1.29 1.03 1.35 Octanol Water Coef. log K (L) -2.48 0.74 -2.24 -2.92 1.13 2.89 2.73 1.93 1.79 1.47 1.81 4.11 1.09 1.15 5.18 3.62 2.36 2.48 2.42 0.60 0.90 2.13 5.91 6.50 4.28 (continued) 4) Use Table 2.3 from your text book to determine the physical state (solid, liquid, or gas) of the following chemicals: Ethane, Ethanol, Hexane, Methane, Methanol, and Naphthalene. Note: Just do not state your answer, show how you determined the physical state of each of these pollutants using relevant physical properties listed in Table 2.3. TABLE 2.3 Some Physical and Chemical Properties of Various Chemicals. Log units represent base 10. Subcooled/Superheated Chemical n-Butane Carbon tetrachloride Chloroform (trichloromethane) 1,1-Dichloroethane 1,2-Dichloroethane Ethane Ethanol Hexane Methane Methylene chloride n-Octane n-Pentane Propane 1.1.1-Trichloroethane (TCA) Trichloroethene (TCE) Vinyl chloride (chloroethene) Aniline Benzene Benz(a)anthracene Benzo(a)pyrene n-Butylbenzene Molecular Weight T To Density (g/mol) (°C) (°C) (g/cm³) 58.1 153.8 119.4 99.0 99.0 Aqueous Vapor Solubility Pressure -log CA -log p (atm) F (mol/L) Aliphatic Compounds and Halogenated Aliphatic Compounds 2.98 2.59 2.20 1.19 1.30 1.07 2.69 30.1 46.0 86.2 16.0 84.9 114.2 72.2 -138.4 -0.5 -22.9 77.0 61.7 -63.5 -97.0 -35.4 -183.3 -114.0 -95.0 -182.5 -164.0 -95.1 -56.8 -129.7 44.1 -189.7 -42.1 133.4 -30.4. 131.4 62.5 57.5 83.5 -88.6 78.0 0.79 69.0 0.66 93.1 -6.3 78.1 5.5 228.3 159.8 252.3 176.5 134.2 -88.0 39.7 125.7 36.1 74.1 -73.0 87.0 -153.8 -13.4 184.0 80.1 435.0 1.59 1.48 1.18 1.24 183.0 1.33 0.70 0.63 1.34 1.46 1.08 0.88 ∞0 3.83 2.82 0.64 5.20 3.25 2.85 Aromatic Compounds 0.41 1.64 7.31 8.22 3.97 0.86 2.07 2.04 1.35 0.82 0.59 0.52 1.04 078 0.69 0.23 1.73 0.16 0.78 1.01 2.89 0.90 9.55 11.14 2.86 Aqueous Vapor Solubility Pressure -log CA -log p (mol/L) 1.09 0.38 1.88 0.76 5.96 6.71 (atm) -0.39 -1.60 -2.44 -0.97 -0.59 Henry's Law Constant log KA (L atm/mol) 8.20 9.63 2.98 1.38 0.60 0.78 0.03 2.69 -0.31 3.14 2.82 0.41 3.47 3.09 2.85 1.29 1.03 1.35 Octanol Water Coef. log K (L) -2.48 0.74 -2.24 -2.92 1.13 2.89 2.73 1.93 1.79 1.47 1.81 4.11 1.09 1.15 5.18 3.62 2.36 2.48 2.42 0.60 0.90 2.13 5.91 6.50 4.28 (continued)
Expert Answer:
Answer rating: 100% (QA)
To solve this question we need to first knowing about the metting point and boiling point of any s... View the full answer

Related Book For 

Operations Management in the Supply Chain Decisions and Cases
ISBN: 978-0073525242
6th edition
Authors: Roger Schroeder, M. Johnny Rungtusanatham, Susan Goldstein
Posted Date:
Students also viewed these accounting questions
-
Ultrasound loudness Assuming no absorption, what is the change in sound intensity level, in dB, in travelling from a position 1.0 cm from a small ultrasound source, to a position 9.0 cm from the...
-
A large manufacturing company produces computer printers that are distributed and sold all over the United States. Due to lack of industry information, the company has a difficult time ascertaining...
-
A large manufacturing company produces computer printers that are distributed and sold all over the United States. Due to lack of industry information, the company has a difficult time ascertaining...
-
Pew Research published survey results from two random samples. Both samples were asked, Have you listened to an audio book in the last year? The results are shown in the table below. a. Find and...
-
Winfrey Designs had an unadjusted credit balance in its Allowance for Doubtful Accounts at December 31, 2014, of $1,800. Required a. Prepare the adjusting entry assuming that Winfrey estimates...
-
Yang Co. is expected to pay a \($21.00\) dividend next year. The dividend will decline by 10 percent annually for the following three years. In Year 5, Yang will sell off assets worth \($100\) per...
-
In an engine working on the diesel cycle, the air-fuel by weight is 50:1. The temperature of air at the beginning of combustion is 40C and the compression ratio is 15. What is the ideal efficiency of...
-
Strite Company issues $2 million, 10-year, 7% bonds at 99, with interest payable on December 31. The straight-line method is used to amortize bond discount. (a) Prepare the journal entry to record...
-
A 7.0 C charge is moving to the right at a speed of 49m/s. A B-field is then induced with a strength of 3.7 T directed out the page. What is the magnitude of the force exerted on the wire? What...
-
In the Chapter 11 Data Analytics Case, you used Tableau to examine a data set and create calculations to compare the relative age of two companies assets. In this case you continue in your role as an...
-
1) The maximum tension an elevator cable can withstand cable can withstand before breaking is 'T'. Assume the mass of the elevator is 'm'. Drawn a free-body diagram of the elevator and use Newton's...
-
Jennifer Davis, D.D.S., opened a dental practice on January 1, 2025. During the first month of operations, the following transactions pccurred. 1. Performed services for patients who had dental plan...
-
Steven Company has fixed costs of $228,650. The unit selling price, variable cost per unit, and contribution margin per unit for the company's two products are as follows: Product Selling Price per...
-
Cherokee Incorporated is a merchandiser that provided the following information: Number of units sold Selling price per unit Variable selling expense per unit Variable administrative expense per unit...
-
Q1. During a race on level ground, Andrea runs with an average velocity of 6.0 m/s to the east, what is Andrea's displacement after 137 s? Q2. Heather and Matthew walk with an average velocity of...
-
Reading 1: Please go to the "Guide to the Employment Standards Act" for Ontario. https:// www.ontario.ca/document/your-guide-employment-standards-act-0 Read the following sections for the pages...
-
Consider the vector field F(x, y, z) = xi+yj+zk. a) Find a function f such that F = Vf and f(0, 0, 0) = 0. f(x, y, z) = b) Use part a) to compute the work done by F on a particle moving along the...
-
Q1) What is the a3 Value Q2) What is the a7 Value Q3) What is the a4 Value Q4) What is the b3 Value Q5) What is the b2 Value Q6) What is the sign of 2nd constraint? A pastry chef at a bakery wants to...
-
A barbershop has been using a level workforce of barbers five days per week, Tuesday through Saturday. The barbers have considerable idle time on Tuesday through Friday, with certain peak periods...
-
A public accounting firm requires the following activities for an audit: a. Draw a network for this project. b. Make a forward pass and a backward pass to determine ES, LS, EF, and LF. c. What are...
-
Management feels there is a seasonal pattern in the above data for the Donut-Hole Shop (see problem 2), with the first two days of a week representing one level; the third and fourth days...
-
In a vapour compression refrigeration system, the condition of refrigerant before entering the compressor is (a) saturated liquid (b) wet vapour (c) dry saturated liquid (d) superheated vapour
-
During a refrigeration cycle, heat is rejected by the refrigerant in (a) compressor (b) condenser (c) evaporator (d) expansion valve
-
The highest temperature during the cycle in vapour compression refrigeration system occurs after (a) compression (b) condensation (c) expansion (d) evaporation

Study smarter with the SolutionInn App


