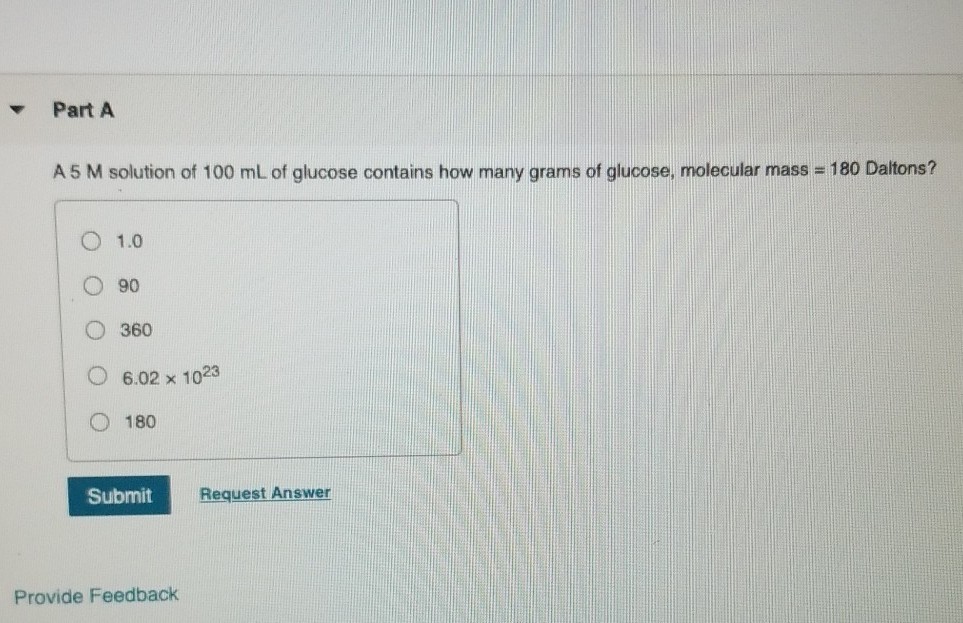
Part A A 5 M solution of 100 mL of glucose contains how many grams of...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Part A A 5 M solution of 100 mL of glucose contains how many grams of glucose, molecular mass = 180 Daltons? 1.0 90 360 6.02 x 1023 180 Submit Provide Feedback Request Answer Part A A 5 M solution of 100 mL of glucose contains how many grams of glucose, molecular mass = 180 Daltons? 1.0 90 360 6.02 x 1023 180 Submit Provide Feedback Request Answer
Expert Answer:
Answer rating: 100% (QA)
Solution The values provided in the question are as follows Molarity of solut... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
A 1.8 m -long wire carries a current of 7.7 A and is immersed within a uniform magnetic field B. When this wire lies along the +x axis, a magnetic force F = (-2.5j) N acts on the wire, and when it...
-
A solution contains 2.0 x 10-4 M Ag+ and 1.5 x 10 -3 M Pb2+ .If NaI is added, will AgI (Ksp = 8.3 x 10-17) or PbI2 (Ksp = 7.9 x 10 -9) precipitate first? Specify the concentration of 1-needed to...
-
A 60.0-mL 0.513 M glucose (C6H12O6) solution is mixed with 120.0 mL of 2.33 M glucose solution. What is the concentration of the final solution? Assume the volumes are additive.
-
Consider the following graph. 2 7 6 C 10 10 H 11 E 12 15 14 (a). Find a minimum spanning tree of the graph using Kruskal's algorithm. List the edges in the order they are put into the tree. (b)....
-
Active Memories Inc. manufactures RAM memory chips for personal computers. An activity analysis was conducted, and the following activity costs were identified with the manufacture and sale of memory...
-
Kitty Wonderland (KW) makes toys for cats and kittens. KWs managers have recently learned that they can calculate the average waiting time for an order from the time an order is received and the time...
-
In the spring of 1999, Source Associates, Inc. (Source), and Conrad A. Mamajek, Inc. (CAM), entered into a joint venture to act as a middleman for the sale of polymers manufactured by Mitsui...
-
Delmott sells a snowboard, Xpert that is popular with snowboard enthusiasts. Below is information relating to Delmotts purchases of Xpert snowboards during September. During the same month, 102 Xpert...
-
Elaborate on why you feel that the chosen concept Tariffs is most important in relation to the wine and spirit industry. How does this affect you as a sales associate working in the wine and spirit...
-
An infinite cylinder of radius R carries a uniform surface charge density o. We propose to set it spinning about its axis, at a final angular velocity ws. How much work will this take, per unit...
-
Calculate the amount of heat released when 3.8 moles of Fe2O3 are formed during the reaction
-
Case Machinery showed the following operating budget for the next year. Complete parts (a) through (d) below. Each scenario is independent. Sales $2,300,000 Fixed cost $1,500,000 Total variable cost...
-
what type of balance sheet account is notes receivable ?
-
why all counties should use the same international accounting standard? pls provide some arguments to support it
-
Based on the the The Malcolm Baldrige National, frame work , choose the company that as been applied that framework and Describe the history of the methodology or framework and how they came to be...
-
What happens when a cardholder approves a transaction?
-
On January 1, 2023 the Bmw Company borrows $60,000 cash by signing a 2-year, 12%, installment note, with quarterly interest payments. 1) Calculate the amount of each payment using the PMT function....
-
One study found that the elderly who do not have children dissave at about the same rate as the elderly who do have children. What might this finding imply about the reason the elderly do not dissave...
-
A complex absorbs red light from a single electron transition. What color is this complex?
-
Concentrated hydrochloric acid contains 1.00 mol HCl dissolved in 3.31 mol H2O. What is the mole fraction of HCl in concentrated hydrochloric acid? What is the molal concentration of HCl?
-
Classify each of the following as a primary, secondary, or tertiary alcohol. a. b. c. d. CH3 CH3 CH3CCH2CHOH CH CH,OH CH,CH2CCH CH3 CH2CH3 CH3CH2COH CH2CH3
-
Construct a frequency and relative frequency histogram of the five-year rate of- return data discussed in Example 3. Approach To draw the frequency histogram, use the frequency distribution in Table...
-
Construct a frequency and relative frequency histogram of the five-year rate of- return data discussed in Example 3. Approach We will use StatCrunch to construct the frequency and relative frequency...
-
The data in Table 14 represent the two-year average percentage of persons living in poverty, by state, for the years 20122013. Draw a stem-and-leaf plot of the data. Approach Step 1 Treat the integer...

Study smarter with the SolutionInn App


