As a technician in a large pharmaceutical research firm, you need to produce 150. mL of...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
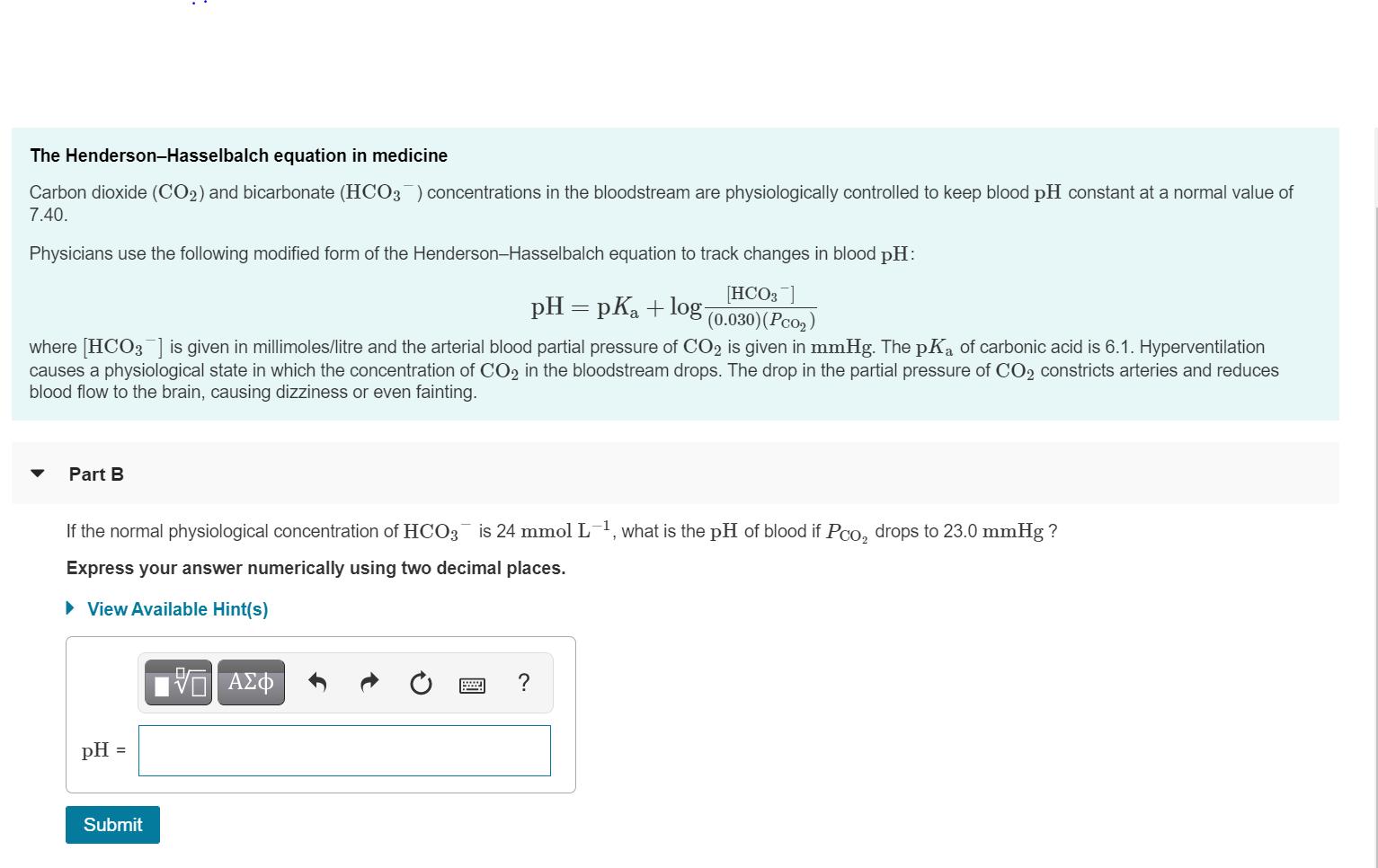
As a technician in a large pharmaceutical research firm, you need to produce 150. mL of 1.00 mol L potassium phosphate buffer solution of pH = 7.09. The pKa of H2PO4 is 7.21. You have the following supplies: 2.00 L of 1.00 mol L KH2PO4 stock solution, 1.50 L of 1.00 mol L K2HPO4 stock solution, and a carboy of pure distilled H20. How much 1.00 mol L KH3PO4 will you need to make this solution? Express your answer to three significant digits with the appropriate units. The Henderson-Hasselbalch equation in medicine Carbon dioxide (CO2) and bicarbonate (HCO3) concentrations in the bloodstream are physiologically controlled to keep blood pH constant at a normal value of 7.40. Physicians use the following modified form of the Henderson-Hasselbalch equation to track changes in blood pH: pH = pKa + log- [HCO3 ] (0.030)(Pco2 where [HCO3] is given in millimoles/litre and the arterial blood partial pressure of CO2 is given in mmHg. The pKa of carbonic acid is 6.1. Hyperventilation causes a physiological state in which the concentration of CO2 in the bloodstream drops. The drop in the partial pressure of CO2 constricts arteries and reduces blood flow to the brain, causing dizziness or even fainting. Part B If the normal physiological concentration of HC03 is 24 mmol L-1, what is the pH of blood if PCo, drops to 23.0 mmHg ? Express your answer numerically using two decimal places. • View Available Hint(s) ΑΣΦ ? pH = Submit As a technician in a large pharmaceutical research firm, you need to produce 150. mL of 1.00 mol L potassium phosphate buffer solution of pH = 7.09. The pKa of H2PO4 is 7.21. You have the following supplies: 2.00 L of 1.00 mol L KH2PO4 stock solution, 1.50 L of 1.00 mol L K2HPO4 stock solution, and a carboy of pure distilled H20. How much 1.00 mol L KH3PO4 will you need to make this solution? Express your answer to three significant digits with the appropriate units. The Henderson-Hasselbalch equation in medicine Carbon dioxide (CO2) and bicarbonate (HCO3) concentrations in the bloodstream are physiologically controlled to keep blood pH constant at a normal value of 7.40. Physicians use the following modified form of the Henderson-Hasselbalch equation to track changes in blood pH: pH = pKa + log- [HCO3 ] (0.030)(Pco2 where [HCO3] is given in millimoles/litre and the arterial blood partial pressure of CO2 is given in mmHg. The pKa of carbonic acid is 6.1. Hyperventilation causes a physiological state in which the concentration of CO2 in the bloodstream drops. The drop in the partial pressure of CO2 constricts arteries and reduces blood flow to the brain, causing dizziness or even fainting. Part B If the normal physiological concentration of HC03 is 24 mmol L-1, what is the pH of blood if PCo, drops to 23.0 mmHg ? Express your answer numerically using two decimal places. • View Available Hint(s) ΑΣΦ ? pH = Submit
Expert Answer:
Answer rating: 100% (QA)
part A The pH of a buffer can be calculated using the Henderson equation which is given below pH ... View the full answer

Related Book For 

Fundamentals of biochemistry Life at the Molecular Level
ISBN: 978-0470547847
4th edition
Authors: Donald Voet, Judith G. Voet, Charlotte W. Pratt
Posted Date:
Students also viewed these physics questions
-
As a technician in a large pharmaceutical research firm, you need to produce 150. mL of 1.00 mol L potassium phosphate buffer solution of pH = 7.09. The pKa of H2PO4 is 7.21. You have the following...
-
You need to produce a set of cylindrical copper wires 350 m long that will have a resistance of 0.125 each. What will be the mass of each of these wires?
-
By mixing available fuel, you need to produce 3500 gallons of gasoline for your feet of trucks this month. You plan to mix 800 gallons of fuel A (80 octane), 1500 gallons of fuel B (92 octane) and...
-
for a manufacturing company product costs include all of the following except direct material overhead costs research and development costs direct labor costs
-
What are the three supports on which market efficiency rests? Why is it that only one of them is required?
-
Refer to the situation described in E 11-25. Alliant prepares its financial statements according to IFRS, and Centerpoint is considered a cash-generating unit. Assume that Centerpoint's fair value of...
-
Use the equation (17.19) suggested by Pai for the turbulent stress and integrate for the velocity profile. How do the results compare with that of Prandtl? = 0.9835 H+ (1-#)['-(1-#)'] (17.19) H+
-
Greener Grass Fertilizer Company plans to sell 200,000 units of finished product in July and anticipates a growth rate in sales of 5 percent per month. The desired monthly ending inventory in units...
-
Create the general journal entries, post to the T - Accounts, and record all applicable entries to the job sheets.a . Raw materials of $ 5 0 , 0 0 0 . 0 0 were purchased with cashb. Factory labor...
-
A residential project has the following information: Four major activities are scheduled across a 4-month time span (shown in the below bar chart). Assuming the monthly indirect cost (i.e., site...
-
what is Wingtech Technology Co. Ltd RECENT ESG Risk Rating. 1) Total ESG Risk Score 2) Environment Risk Score 3) Social Risk Score 4) Government Risk Score
-
How will the selected BPM approach impact the likelihood of success?
-
Why is sustainability important?
-
What is the purpose of communication at this stage of the BPM activity?
-
What are the five levels of a process asset structure? Provide a brief description of each level.
-
The diagram depicted in Figure 1.39 is an automatic closed-loop system for paper moisture level control. Desired Moisture Controller Drier moisture level level Moisture meter Figure 1.39 A...
-
Convert the following decimal numbers to 8-bit two's complement binary numbers and carry out the additions in binary. Indicate whether the sum overflows the 8-bit result. If not show the result as a...
-
Assume a simple Keynesian depression economy with a multiplier of 4 and an initial equilibrium income of $3,000. Saving and investment equal $400, and assume full employment income is $4,000. a. What...
-
The half-cell reduction potential is provided by the Nernst equation (Equation 14-8): (a) On the graph below, plot the reduction potentials for the FADH2/FAD half-cell (E°² = - 0.219 V) when...
-
Some mitochondria use a second codon, in addition to AUG, to specify Met. Which codon(s) is(are) most likely to be used this way?
-
Broken DNA can be repaired by DNA ligase (single-strand breaks) or nonhomologous end-joining (double-strand breaks). Explain why the cell's set of repair enzymes also includes tyrosyl-DNA...
-
Give an example of a requirement on a computer printer.
-
Briefly describe the distinction between requirements and specification.
-
Give an example of a requirement on a digital still camera.

Study smarter with the SolutionInn App