PCC in CHCl H3PO4, A 1) PhCO3H in CHCl2 2) CH3ONA in CH3OH O3 in CHCl2...
Fantastic news! We've Found the answer you've been seeking!
Question:
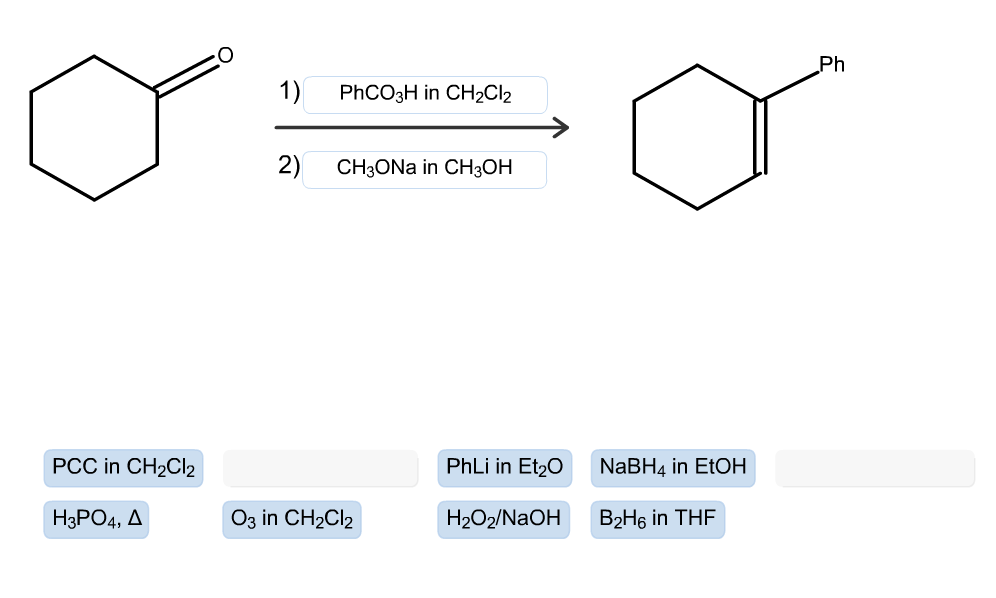
Transcribed Image Text:
PCC in CH₂Cl₂ H3PO4, A 1) PhCO3H in CH₂Cl2 2) CH3ONA in CH3OH O3 in CH₂Cl2 PhLi in Et₂O H₂O2/NaOH NaBH4 in EtOH B₂H6 in THF Ph PCC in CH₂Cl₂ H3PO4, A 1) PhCO3H in CH₂Cl2 2) CH3ONA in CH3OH O3 in CH₂Cl2 PhLi in Et₂O H₂O2/NaOH NaBH4 in EtOH B₂H6 in THF Ph
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A mixture of 2 mol of CO2 and 1 mol of O2 is heated to 3200 K at a pressure of 2 atm. Determine the equilibrium composition of the mixture, assuming that only CO2, CO, O2, and O are present.
-
A mixture of 1 mol of H2O, 2 mol of O2, and 5 mol of N2 is heated to 2200 K at a pressure of 5 atm. Assuming the equilibrium mixture consists of H2O, O2, N2, and H2, determine the equilibrium...
-
In Exercises 1 and 2, determine if b is a linear combination of a1, a2, and a3. 1. 2. 216 120 675 101
-
Consider the real projective n-space RP" obtained by identifying each point S" with its antipode -r. (a) Compute its fundamental group. (b) Find all its covering spaces up to isomorphism.
-
Discuss some types of conflict that might arise during a project. Describe two situations in which you have experienced these types of conflict.
-
David Collins died during the current year. The personal representative of Davids estate reviewed the following assets: Stocks in Davids name...
-
Suppose Sports-R-Us purchases $40,000 of sportswear on account from Pacific Trail on March 1,2010. Credit terms are 2/10, net 30. Sports-R-Us pays Pacific Trail on March 8, 2010. 1. Journalize the...
-
Michael Jacks deposited $500,000 into a bank for 6 months. At the end of that time, he withdrew the money and received $520,000. If the bank paid interest based on continuous compounding: (a) What...
-
1. Need to have at least 3 out of 5 attributes better than the benchmark (row 26, Cells D-I), duration can be longer if you feel that the yields will drop 2. A minimum of 12 ETFS need to be used to...
-
Upsidedown Cake Company produces dessert products for sale in grocery stores, but it also has a retail location. At the end of 2018, the company had $349,000 in accounts receivable before netting out...
-
Hope is a marketing manager at a local company. Information about her 2019 income and expenses is as follows: Income received Salary $150,000 Taxes withheld from salary: Federal income tax $30,000...
-
What kind of impact do you think one company switching to less environmentally damaging practices could have on the general atmosphere of the shipping world?
-
What is the standard index? What is the best-fit index?
-
If you were told that you would take a personal pay cut from switching to the more environmentally friendly route, how would that affect your decision?
-
The last step in the selling process is closing the sale. True or False
-
Using marketing intermediaries often adds efficiency to the supply chain. True or False
-
Suppose you had a 0.5 in diameter silver antenna that was exposed to the Venus surface atmosphere at 864F for 1 minute. Assuming the film transfer coefficient, h = 9 Btu/(hr. ft2 F) and the antenna...
-
Design and describe an application-level protocol to be used between an automatic teller machine and a bank's centralized computer. Your protocol should allow a user 's card and password to be...
-
Outline a synthesis of the quaternary ammonium salt (CH 3 ) 3 Br from each of the following combinations of starting materials. Benzylamine and any other reagents NCH,Ph
-
Outline laboratory syntheses of each of the following compounds, starting with benzene and any other reagents. (The references to equations will assist you with nomenclature.) (a) p-dibromobenzene...
-
The following natural product readily gives a Diels-Alder adduct with maleic anhydride (structure in Eq. 15.12a, p. 698) under mild conditions. What is the most likely configuration of the two double...
-
Which one of the following is an exact straight line mechanism using lower pairs? (a) Watt's mechanism (b) Grasshopper mechanism (c) Robert's mechanism (d) Peaucellier's mechanism.
-
In the epicyclic gear train shown in the Fig. \(88, T_{A}=40, T_{B}=20\). For three revolutions of the arm, the gear \(B\) will rotate through (a) 6 revolutions (b) 2.5 revolutions (c) 3 revolutions...
-
Which one of the following statements in respect of involute profiles for gear teeth is not correct? (a) Interference occurs in involute profiles (b) Involute tooth form is sensitive to change in...

Study smarter with the SolutionInn App



