Question: Your enzyme is inhibited by one of the compounds shown in the corresponding box PDB code : 2V1X, residue position (46), mutation( PHE) 1CJY inhibitor
Your enzyme is inhibited by one of the compounds shown in the corresponding box
PDB code : 2V1X, residue position (46), mutation( PHE)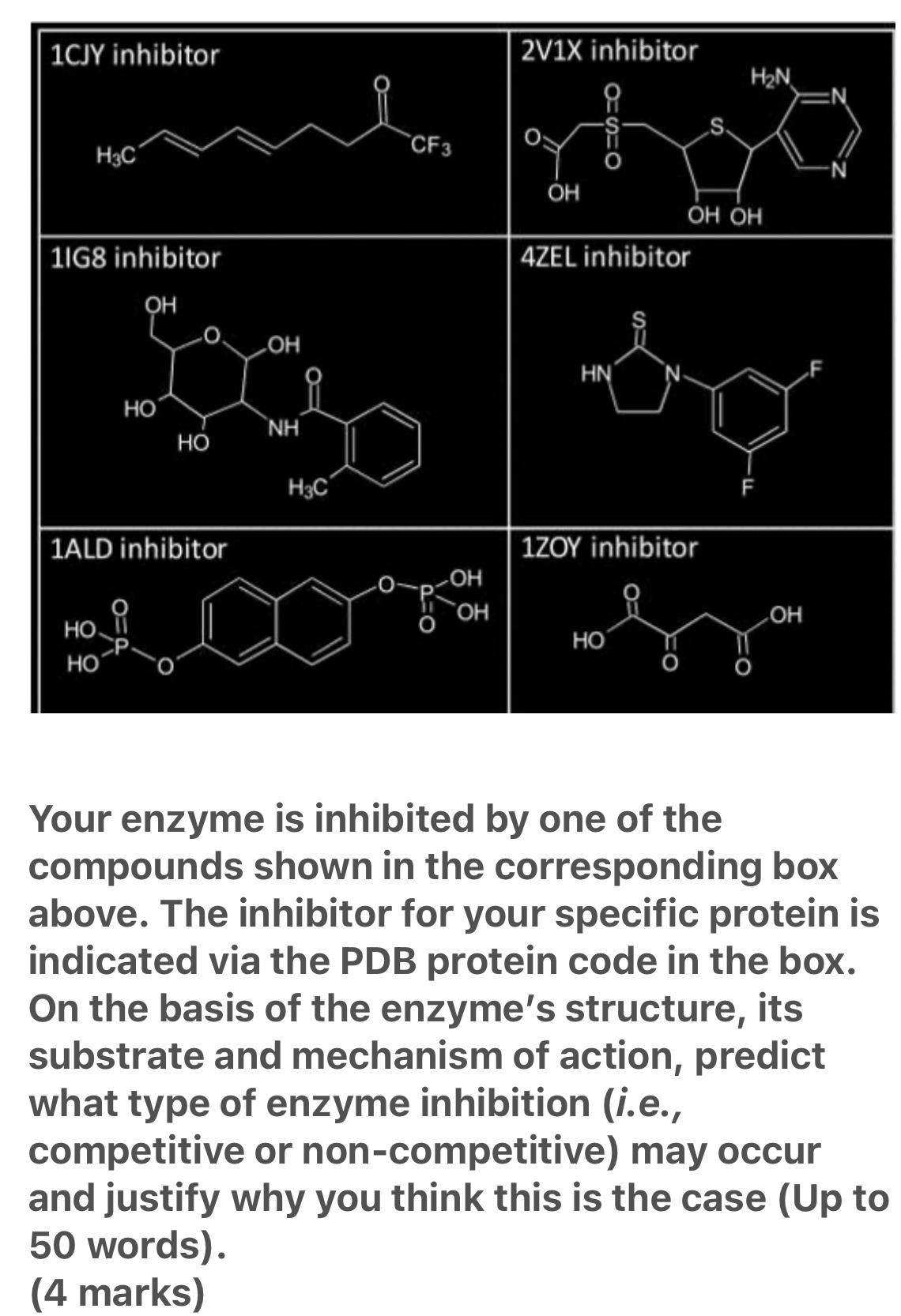
1CJY inhibitor 2V1X inhibitor H2N .S. CF3 H3C OH 1IG8 inhibitor 4ZEL inhibitor OH LOH HN HO NH HO H3C 1ALD inhibitor 1ZOY inhibitor HO . HO HO Your enzyme is inhibited by one of the compounds shown in the corresponding box above. The inhibitor for your specific protein is indicated via the PDB protein code in the box. On the basis of the enzyme's structure, substrate and mechanism of action, predict what type of enzyme inhibition (i.e., competitive or non-competitive) may occur and justify why you think this is the case (Up to 50 words). (4 marks) its
Step by Step Solution
3.37 Rating (166 Votes )
There are 3 Steps involved in it
A limited enzyme has been identified as yexokinase pII Hexokinase is the primary glycolytic enzyme w... View full answer

Get step-by-step solutions from verified subject matter experts


