Percentage of radioactive isotope remaining 100 90 80 70 60 8 6 8 8 8 20...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
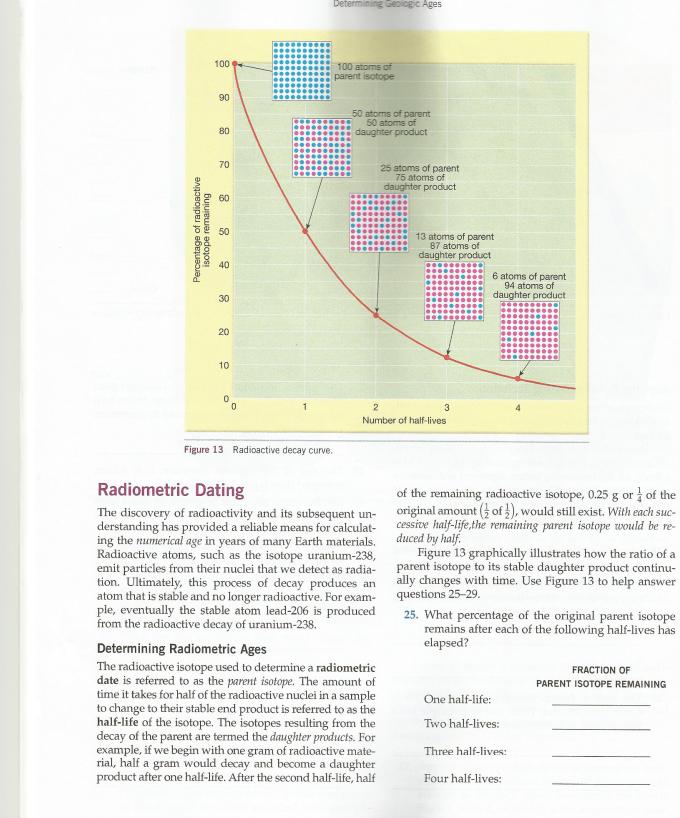
Percentage of radioactive isotope remaining 100 90 80 70 60 8 6 8 8 8 20 10 0 1 Determining Geologic Ages 100 atoms of parent isotope Figure 13 Radioactive decay curve. 50 atoms of parent 50 atoms of daughter product Radiometric Dating The discovery of radioactivity and its subsequent un- derstanding has provided a reliable means for calculat- ing the numerical age in years of many Earth materials. Radioactive atoms, such as the isotope uranium-238, emit particles from their nuclei that we detect as radia- tion. Ultimately, this process of decay produces an atom that is stable and no longer radioactive. For exam- ple, eventually the stable atom lead-206 is produced from the radioactive decay of uranium-238. 25 atoms of parent 75 atoms of daughter product 2 Number of half-lives Determining Radiometric Ages The radioactive isotope used to determine a radiometric date is referred to as the parent isotope. The amount of time it takes for half of the radioactive nuclei in a sample to change to their stable end product is referred to as the half-life of the isotope. The isotopes resulting from the decay of the parent are termed the daughter products. For example, if we begin with one gram of radioactive mate- rial, half a gram would decay and become a daughter product after one half-life. After the second half-life, half 13 atoms of parent 87 atoms of daughter product 3 6 atoms of parent 94 atoms of daughter product of the remaining radioactive isotope, 0.25 g or of the original amount (of), would still exist. With each suc- cessive half-life,the remaining parent isotope would be re- duced by half. Figure 13 graphically illustrates how the ratio of a parent isotope to its stable daughter product continu- ally changes with time. Use Figure 13 to help answer questions 25-29. 25. What percentage of the original parent isotope remains after each of the following half-lives has elapsed? One half-life: Two half-lives: Three half-lives: Four half-lives: FRACTION OF PARENT ISOTOPE REMAINING Percentage of radioactive isotope remaining 100 90 80 70 60 8 6 8 8 8 20 10 0 1 Determining Geologic Ages 100 atoms of parent isotope Figure 13 Radioactive decay curve. 50 atoms of parent 50 atoms of daughter product Radiometric Dating The discovery of radioactivity and its subsequent un- derstanding has provided a reliable means for calculat- ing the numerical age in years of many Earth materials. Radioactive atoms, such as the isotope uranium-238, emit particles from their nuclei that we detect as radia- tion. Ultimately, this process of decay produces an atom that is stable and no longer radioactive. For exam- ple, eventually the stable atom lead-206 is produced from the radioactive decay of uranium-238. 25 atoms of parent 75 atoms of daughter product 2 Number of half-lives Determining Radiometric Ages The radioactive isotope used to determine a radiometric date is referred to as the parent isotope. The amount of time it takes for half of the radioactive nuclei in a sample to change to their stable end product is referred to as the half-life of the isotope. The isotopes resulting from the decay of the parent are termed the daughter products. For example, if we begin with one gram of radioactive mate- rial, half a gram would decay and become a daughter product after one half-life. After the second half-life, half 13 atoms of parent 87 atoms of daughter product 3 6 atoms of parent 94 atoms of daughter product of the remaining radioactive isotope, 0.25 g or of the original amount (of), would still exist. With each suc- cessive half-life,the remaining parent isotope would be re- duced by half. Figure 13 graphically illustrates how the ratio of a parent isotope to its stable daughter product continu- ally changes with time. Use Figure 13 to help answer questions 25-29. 25. What percentage of the original parent isotope remains after each of the following half-lives has elapsed? One half-life: Two half-lives: Three half-lives: Four half-lives: FRACTION OF PARENT ISOTOPE REMAINING
Expert Answer:
Answer rating: 100% (QA)
what percentage of the original parent isotope remains after each of the following halflives has ela... View the full answer

Related Book For 

Introduction to Statistical Investigations
ISBN: 978-1118922002
1st edition
Authors: Beth L.Chance, George W.Cobb, Allan J.Rossman Nathan Tintle, Todd Swanson Soma Roy
Posted Date:
Students also viewed these american history questions
-
Amog has been following the stock market for a while and is particularly interested in investing in Godrej. The stock price of the company is currently volatile and he wants to try and make some...
-
The accompanying diagram shows the demand, marginal revenue, and marginal cost of a monopolist.a. Determine the profit-maximizing output and price.b. What price and output would prevail if this...
-
Consider the competitive market for steel. Assume that, regardless of how many firms are in the industry, every firm in the industry is identical and faces the marginal cost (MC), average total cost...
-
3. Six Sigma College of Business offers five different courses of study. The planned enrollment in each course is 25 students. Registration for the upcoming semester of study has been recently...
-
1. Why does the court criticize MPS for recognizing the out-of-state union as the exclusive bargaining representative of the workers? 2. Suppose that MPS did recognize the local union and,...
-
Trade secrets do not have to be novel, be non-obvious, or have utility. If that is the case, then what is the economic advantage to a trade secret?
-
The [+45/-45] laminate described in Problem 7.6 is subjected to a uniaxial force per unit length \(N_{x}=30 \mathrm{MPa} \mathrm{mm}\). Find the resulting stresses and strains in each ply along the...
-
Distinguish a significant deficiency in internal control from a material weakness in internal control. How will the presence of one significant deficiency affect an auditor's report on internal...
-
Suzy was killed on a rainy hot summer night. Her business partner, Don, waited behind a muddy dirt hill and shot Suzy as she was unlocking her home entrance door. The District Attorney charged Don...
-
4.9 Estimate the static formation temperature from the following data (from Ref. 30): depth=7,646 ft, drilling stopped-22:00
-
1.9.3 Show that, for an exponential survival distribution, the probability of surviving past time t + t given that an individual has survived past to equals the unconditional probability of surviving...
-
On the human factors and human engineering issues in user interface design. The spirt of the discussion is that it is imperative that we understand the people who will be using the system and that we...
-
The only two stores in a mall decide whether to hire one guard or none-extra guards provide no extra protection. The guard patrolling the mall provides a service without rivalry, simultaneously...
-
Skousen Exploration Corporation was formed on January 1, 20X3. The company was formed by Cliff and Chris Skousen with the goal of conducting geophysical support services related to natural gas...
-
Hamelin Hospital is a large (700-bed) regional hospital in the northeastern United States. The information technology (IT) department employs 75 people and has an operating budget of over $35...
-
Bingo Corporation is a newly formed company. Below are the first 10 transactions that Bingo encountered. Prepare an income statement, statement of retained earnings, and balance sheet immediately...
-
Evan's team had just launched their new app. They held a big party that night for the whole staff. It was in a nightclub with an open bar. Evan began the night mingling and avoided alcohol. But,...
-
Cassandra Casey operates the Futuristic Antique Store. She maintains subsidiary ledgers for accounts payable and accounts receivable. She presents you with the following information for October 2019:...
-
Two researchers want to investigate whether there is a relationship between annual company profit ($) and median annual salary paid by the company ($). Researcher Bart collects data on a random...
-
A research article, Power Posing: Brief Nonverbal Displays Aff ect Neuroendocrine Levels and Risk Tolerance, published in Psychological Science, September 2010, describes a study involving 42...
-
Refer to Exercise 2.CE.9. a. Based on the test results found in Exercise 2.CE.9, part (b), if you are testing at a significance level of 5% and you have made an error, what type of error have you...
-
After successful stretches at Target and Apple, it seemed as though Ron Johnson was a master marketer. But things went sour quickly after JC Penney hired him as its CEO. Johnsons attempt to reinvent...
-
Sharee Garza developed a new type of space heater that is quieter and safer than previous generations of space heaters. People living in small spaces, such as apartments or dorm rooms, are the target...
-
In 1995, two dermatologists, Dr. Katie Rodin and Dr. Kathy Fields, developed what they believed was a medical breakthrough to fight acne. Their mission: to help millions of people rid themselves of...

Study smarter with the SolutionInn App


