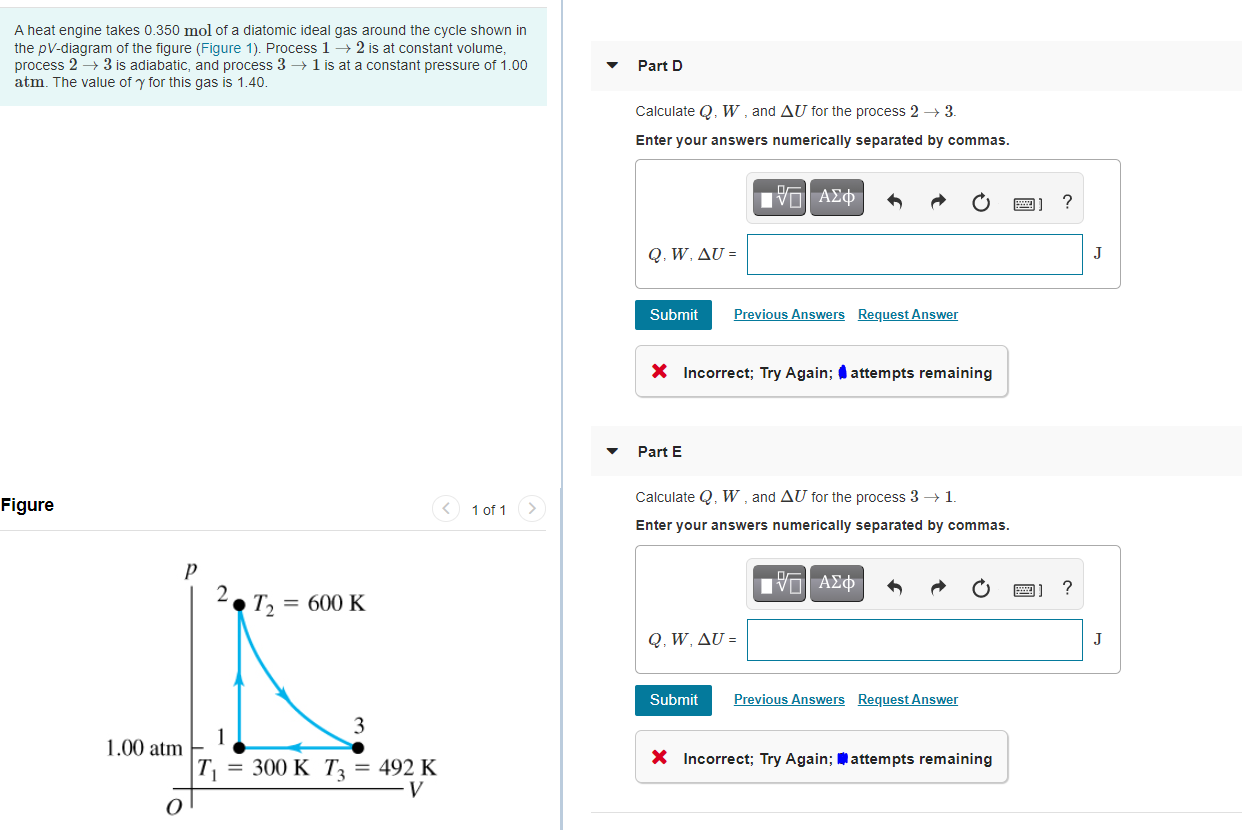
A heat engine takes 0.350 mol of a diatomic ideal gas around the cycle shown in...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A heat engine takes 0.350 mol of a diatomic ideal gas around the cycle shown in the pV-diagram of the figure (Figure 1). Process 1– 2 is at constant volume, process 2 → 3 is adiabatic, and process 3 → 1 is at a constant pressure of 1.00 atm. The value of y for this gas is 1.40. Part D Calculate Q. W, and AU for the process 2 → 3. Enter your answers numerically separated by commas. ? Q. W. ΔU = J Submit Previous Answers Request Answer X Incorrect; Try Again; attempts remaining Part E Calculate Q, W , and AU for the process 3 → 1. Figure 1 of 1 Enter your answers numerically separated by commas. Ηνα ΑΣφ ? 2. T2 = 600 K Q, W. ΔU - J Submit Previous Answers Request Answer 3 1 1.00 atm X Incorrect; Try Again; attempts remaining T = 300 K T; = 492 K A heat engine takes 0.350 mol of a diatomic ideal gas around the cycle shown in the pV-diagram of the figure (Figure 1). Process 1– 2 is at constant volume, process 2 → 3 is adiabatic, and process 3 → 1 is at a constant pressure of 1.00 atm. The value of y for this gas is 1.40. Part D Calculate Q. W, and AU for the process 2 → 3. Enter your answers numerically separated by commas. ? Q. W. ΔU = J Submit Previous Answers Request Answer X Incorrect; Try Again; attempts remaining Part E Calculate Q, W , and AU for the process 3 → 1. Figure 1 of 1 Enter your answers numerically separated by commas. Ηνα ΑΣφ ? 2. T2 = 600 K Q, W. ΔU - J Submit Previous Answers Request Answer 3 1 1.00 atm X Incorrect; Try Again; attempts remaining T = 300 K T; = 492 K
Expert Answer:

Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Posted Date:
Students also viewed these physics questions
-
A beat engine takes 0.350 mol of a diatomic ideal gas around the cycle shown in the pV-diagram of fig. 20.24. Process I 2 is at constant volume, process 23 is adiabatic, and process 3 1 is at a...
-
A monatomic ideal gas is taken around the cycle shown in Fig. 20.29 in the direction shown in the figure. The path for process c → a is a straight line in the pV-diagram. (a) Calculate Q, W, and...
-
In Figure where V23 = 3.00V1, n moles of a diatomic ideal gas are taken through the cycle with the molecules rotating but not oscillating. What are? (a) p2/p1 (b) p3/p1 and (c) T3/T1 For path 1 ?? 2,...
-
A company is implementing Dynamics 365 Supply Chain Management. The company plans to implement the solution in a phased approach across several incremental projects. The project manager needs a...
-
Key comparative figures for both Apple and Google follow. Required 1. Compute the dollar amount of gross margin and the gross margin ratio for the two years shown for each of these companies. 2....
-
What are some of the innovative activities performed by Amazon at their fulfillment centers?
-
For accounting purposes, how are liabilities defined?
-
Lotus Fixtures, Inc. (LFI), manufactures steel fittings. Each fitting requires both steel and an alloy that allows the fitting to be used under extreme conditions. The following data apply to the...
-
Richland Crane ( U . S . ) exports heavy crane equipment to several Chinese dock facilities. Sales are currently 1 0 comma 0 0 0 units per year at the yuan equivalent of USD 2 6 comma 0 0 0 each. The...
-
Consider the blending system shown in Figure. A feedback control system is used to reduce the effect of disturbances in feed composition, x 1 , n the controlled variable, product composition, x....
-
How does the adoption of International Financial Reporting Standards (IFRS) affect the consolidation process for a multinational corporation? And how does it impact the presentation of the...
-
Choose the main ownership type that you feel would be best to use if you decided to start and operate your own business.(partnership, sole proprietor, etc.) You can talk about a real business that...
-
Presented here are the comparative balance sheets of Hames Incorporated at December 31, 2023 and 2022. Sales for the year ended December 31, 2023, totaled $590,000. Check m HAMES INCORPORATED Balance...
-
A company is considering two mutually exclusive expansion plans. Plan A requires a $40 million expenditure on a large-scale integrated plant that would provide expected cash flows of $6.39 million...
-
How does the intricate interplay between organizational culture, stakeholder engagement, and strategic alignment serve as the cornerstone for efficacious Change Management initiatives within dynamic...
-
Cost Behavior Alisha Incorporated manufactures medical stents for use in heart bypass surgery. Based on past experience, Alisha has found that its total maintenance costs can be represented by the...
-
1.If you were an entrepreneur, how would you think of the difference between a preferred-plus-cheap common structure and a convertible preferred structure?
-
On 1 July 2018, Parent Ltd acquired all the shares of Son Ltd, on a cum-div. basis, for $2,057,000. At this date, the equity of Son Ltd consisted of: $ 1,000,000 Share capital 500 000 shares...
-
How much energy is released when a 238U nucleus decays by emitting? (a) An alpha particle and (b) A sequence of neutron, proton, neutron, proton? (c) Convince yourself both by reasoned argument and...
-
In an experiment, a rectangular block with height h is allowed to float in four separate liquids. In the first liquid, which is water, it floats fully submerged. In liquids A, B, and C, it floats...
-
Earth has a mass of 5.98 x 1.0 24 kg. The average mass of the atoms that make up Earth is 40 u. How many atoms are there in Earth?
-
In order to create a digital signature a. the sender encrypts a hash using the recipients public key b. the sender encrypts a hash using the senders private key c. the sender encrypts a hash using...
-
Explain how each of the following security procedures can increase system reliability: 1 Encryption 2 Employee security awareness training 3 Eirewalls 4 IDS 5 VPNs
-
Modifying default configurations to improve security is called a. encryption hardening b. patching dialing c. hardening d. war dialing

Study smarter with the SolutionInn App


