A watch's luminous glow is due to zinc sulfide paint that is energized by beta particles...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
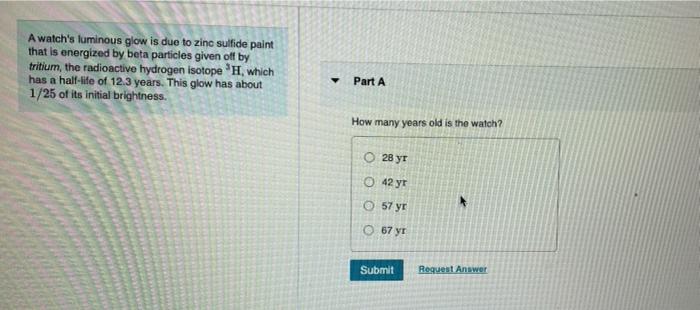
A watch's luminous glow is due to zinc sulfide paint that is energized by beta particles given off by tritium, the radioactive hydrogen isotope H, which has a half-life of 12.3 years. This glow has about 1/25 of its initial brightness. Part A How many years old is the watch? O 28 yr O 42 yr O 57 yr O 67 yr Submit Request Answer A watch's luminous glow is due to zinc sulfide paint that is energized by beta particles given off by tritium, the radioactive hydrogen isotope H, which has a half-life of 12.3 years. This glow has about 1/25 of its initial brightness. Part A How many years old is the watch? O 28 yr O 42 yr O 57 yr O 67 yr Submit Request Answer
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
The radioactive hydrogen isotope 3 H, called tritium, has a half-life of 12 years. a. What are the decay mode and the daughter nucleus of tritium? b. What are the lifetime and the decay rate of...
-
Tritium (1H3) is a rare isotope of hydrogen that can be produced by the fusion reaction ZX1 + 1Y 1H3 + Y The atomic messes are ZX1 (1.0087), 1 YA (2.0141u), 1H3(3.0161u). Determine the (a) atomic...
-
Hydrogen sulfide is composed of two elements: hydrogen and sulfur. In an experiment, 6.500 g of hydrogen sulfide is fully decomposed into its elements. (a) If 0.384 g of hydrogen is obtained in this...
-
Given the monthly returns that follow, find the R2, alpha, and beta of the portfolio. Compute the average return differential with and without sign. Do not round intermediate calculations. Round your...
-
Pharmacists at Wal-Mart were paid a salary based on an expected number of hours of work. During slow times of the year, pharmacists hours and salaries were reduced in response to lower sales. The...
-
Soil borings made at a site near Chicago indicate that the top 6 m is a loose sand and miscellaneous fill, with the groundwater table at 3 m below the ground surface. Below this is a fairly soft...
-
A capacitor is attached to an AC voltage source. Which change will result in a doubling of the current? A. Halving the voltage and doubling the frequency B. Doubling the frequency C. Halving the...
-
Consider the data for McConnell Department Stores presented in P15-23A. Requirements 1. Prepare a common-size income statement and balance sheet for McConnell. The first column of each statement...
-
From the following, prepare a balance sheet for Kim Co. Cleaners at the end of September 202X: Cash, $67,000; Equipment, $7,000; Accounts Payable, $13,000; B. Kim, Capital. Prepare the balance sheet....
-
Ellery Products manufactures various components for the fashion industry. Ellery buys fabric from two vendors: Ewers Textiles and Bramford Materials. Ellery chooses the vendor based on price. Once...
-
You are an advisor to a woman who wants to turn her hobby of making sourdough break into a viable business. Give her a description of the decisions she needs to make during each stage of the...
-
On 1 January 20x1, ACE Corporation introduced a share appreciation rights (SARs) plan for 20 selected senior managers. Under the plan, each manager was granted 10,000 share appreciation rights, which...
-
Star Corporation is a provider of computer software and IT services in two large regions of Easter Europe. The company uses 12 percent to evaluate investments; however, due to latest developments it...
-
Imagine that you were a new CFO of Beily Inc., a childrens bicycle manufacturer. The president, Mr. Zhao, started the business 2 years ago. The firm manufactures two types of products, bicycles for...
-
Top Performance has been operating for several years in the outskirts of Ostersund, Sweden, and is a new manufacturer of a top-of-the-line outdoor jacket. You are starting an internship as assistant...
-
The debt and equity section of the TOTAL Group balance sheet is shown here (in million euros). The current market price of the common shares is 200. Reconstruct the financial statement assuming that...
-
9. A time-study analyst timed an assembly task for 30 cycles, and then computed the average time per cycle, which was 18.75 minutes. The analyst assigned a performance rating of 0.96 to the operator...
-
Solve the relation Exz:Solve therelation ne %3D
-
Crystalline silicon has a cubic structure. The unit cell edge length is 543 pm. The density of the solid is 2.33 g/cm3. Calculate the number of Si atoms in one unit cell?
-
The 235U isotope undergoes fission when bombarded with neutrons. However, its natural abundance is only 0.72 percent. To separate it from the more abundant 238 U isotope, uranium is first converted...
-
Cysteine, shown here, is one of the 20 amino acids found in proteins in humans. Write the molecular formula and calculate its percent composition by mass.
-
In Appendix 16A.1, we illustrate the calculation of a standard error for the marginal effect in a probit model of transportation, Example 16.4. In the appendix, the calculation is for the marginal...
-
In Example 16.3, we illustrate the calculation of the likelihood function for the probit model in a small example. a. Calculate the probability that \(y=1\) if \(x=1.5\), given the values of the...
-
In Examples 16.2 and 16.4, we presented the linear probability and probit model estimates using an example of transportation choice. The logit model for the same example is \(P(A U T...

Study smarter with the SolutionInn App


