Question: A system absorbs 100 KJ heat in the process shown in the figure. What is AU for the system? PA 2 bar 1 bar
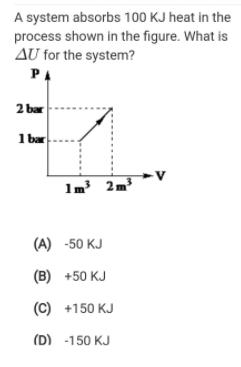
A system absorbs 100 KJ heat in the process shown in the figure. What is AU for the system? PA 2 bar 1 bar 1m' 2m (A) -50 KJ (B) +50 KJ (C) +150 KJ (D) -150 KJ
Step by Step Solution
3.49 Rating (149 Votes )
There are 3 Steps involved in it
Answe... View full answer

Get step-by-step solutions from verified subject matter experts


