Question: What is the value (in V) of Ecell for the following reaction? Co+ (aq) + Fe (s) Co (s) + Fe+ (aq) Question 3
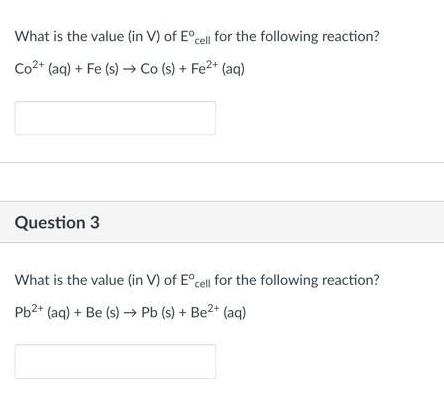
What is the value (in V) of Ecell for the following reaction? Co+ (aq) + Fe (s) Co (s) + Fe+ (aq) Question 3 What is the value (in V) of Ecell for the following reaction? Pb+ (aq) + Be (s) Pb (s) + Be+ (aq)
Step by Step Solution
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


