At 30.0C, the molar solubility of barium sulfate in water is 1.20x10-5 M. Calculate the solubility...
Fantastic news! We've Found the answer you've been seeking!
Question:
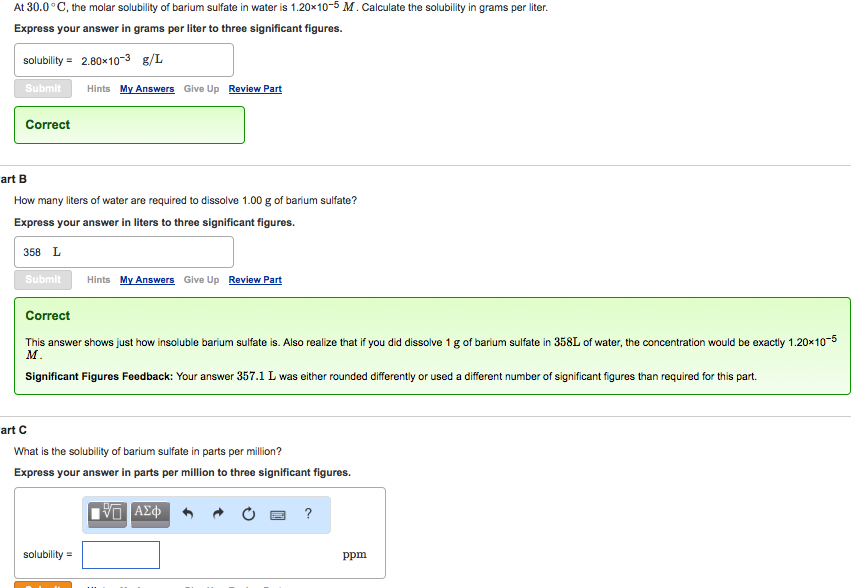
Transcribed Image Text:
At 30.0°C, the molar solubility of barium sulfate in water is 1.20x10-5 M. Calculate the solubility in grams per liter. Express your answer in grams per liter to three significant figures. solubility = 2.80×10-3 g/L Submit Correct Hints My Answers Give Up Review Part art B How many liters of water are required to dissolve 1.00 g of barium sulfate? Express your answer in liters to three significant figures. 358 L Submit Hints My Answers Give Up Review Part Correct This answer shows just how insoluble barium sulfate is. Also realize that if you did dissolve 1 g of barium sulfate in 358L of water, the concentration would be exactly 1.20x10-5 M. Significant Figures Feedback: Your answer 357.1 L was either rounded differently or used a different number of significant figures than required for this part. solubility= art C What is the solubility of barium sulfate in parts per million? Express your answer in parts per million to three significant figures. ΨΠ ΑΣΦ ? ppm At 30.0°C, the molar solubility of barium sulfate in water is 1.20x10-5 M. Calculate the solubility in grams per liter. Express your answer in grams per liter to three significant figures. solubility = 2.80×10-3 g/L Submit Correct Hints My Answers Give Up Review Part art B How many liters of water are required to dissolve 1.00 g of barium sulfate? Express your answer in liters to three significant figures. 358 L Submit Hints My Answers Give Up Review Part Correct This answer shows just how insoluble barium sulfate is. Also realize that if you did dissolve 1 g of barium sulfate in 358L of water, the concentration would be exactly 1.20x10-5 M. Significant Figures Feedback: Your answer 357.1 L was either rounded differently or used a different number of significant figures than required for this part. solubility= art C What is the solubility of barium sulfate in parts per million? Express your answer in parts per million to three significant figures. ΨΠ ΑΣΦ ? ppm
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Please help me with this its urgent please help me for freee How will the social sciences affect your life in the following areas and explain why: Area of Life Effect and why Work / Career Home and...
-
Can someone please help me with correcting this resonance?Please help me to draw the resonance structures on CHEMDRAW and putthe correct arrows. Can someone also please help me with the resonance...
-
14. The basis for recording direct and indirect labor costs incurred is a summary of the period's: a. job order cost sheets b. time tickets employees' earnings records C. d. clock cards 15. At the...
-
The par yield curve for U.S. Treasury bonds is currently flat across all maturities at 5.50 percent. You have observed following "paired" transaction by your bond portfolio manager: Bond G H...
-
What is an evaluation model? How would you create a weighted evaluation model?
-
The Molokai Nut Company (MNC) makes four different products from macadamia nuts grown in the Hawaiian Islands: chocolate-coated whole nuts (Whole), chocolate-coated nut clusters (Cluster),...
-
The following data are for Marvin Department Store. The account balances (in thousands) are for 2017. 1. Compute (a) the cost of goods purchased and (b) the cost of goods sold. 2. Prepare the income...
-
Based on the information below, calculate the weighted average cost of capital. Great Corporation has the following capital situation. Debt: One thousand bonds were issued five years ago at a coupon...
-
6. (a) Given == 2-23i.Find |=| and arg (=*). Hence, solve z=2-23i leave your answers in polar form. (b) Given that z = 1 + i is a root of the polynomial equation z4-4z3 +11z2 - 14z + 10 = 0. Find the...
-
You are valuing Melissa Corporation. You have discounted the future free cash flows back to the present at an appropriate discount rate and get a present value of $140,000,000. When reviewing the...
-
Trevor was looking to purchase a vehicle off of Craigslist. He found a vehicle he really liked but prior to completing the purchase, he did a title search to make sure there were no leans against the...
-
(a) Suppose that your professor asks you to compare China and India based on income inequality. Which measure would you use and explain how would you determine which country is doing better based on...
-
Employees recently elected a new Health and Safety Representative (HSR) and your HR Manager has asked you to ensure that the HSR is trained and ready to undertake the responsibilities of the role. As...
-
2. Short answer A. What are the key factors influencing the choice between debt or equity financ- ing for a new business? B. Describe why "monitoring" is a crucial element of successful tacit...
-
In a population of 300,000 people, 120,000 are infected with a virus. After a person becomes infected and then recovers, the person is immune (cannot become infected again). Of the people who are...
-
10. In a paper published in the Proceedings of the National Academy of Sciences of the United States of America (PNAS) entitled "Evidence on the impact of sustained exposure to air pollution on life...
-
On December 31, Reggit Company held the following short-term investments in its portfolio of available-for-sale debt securities. Reggit had no short-term investments in its prior accounting periods....
-
Why did management adopt the new plan even though it provides a smaller expected number of exposures than the original plan recommended by the original linear programming model?
-
A gas in a container had a measured pressure of 57 kPa. Calculate the pressure in units of atm and mmHg.
-
Ethanolamine, HOC2H4NH2, is a viscous liquid with an ammonialike odor; it is used to remove hydrogen sulfide from natural gas. A 0.15 Maqueous solution of ethanolamine has a pH of 11.34. What is Kb...
-
The reaction 3A(g) +B(s) 2C(aq) + D(aq) occurs at 25C in a flask, which has 1.87 L available for gas. After the reaction attains equilibrium, the amounts (mol) or concentrations (M) of substances...
-
Given your response to item 2, how would these measures be useful? What could they be used for? Turnover is costly for organizations. In addition to the direct costs of recruiting, hiring, and...
-
How does New Belgium introduce fun into the workplace? How does having fun keep employees engaged and enthusiastic about their work and the company? Kim Jordan and Jeff Lebesch, the husband and wife...
-
Visit the New Belgium Brewing Web site (www.newbelgium.com) to learn more about the company. The company has a strong culture that values environmental sustainability. Here are its core beliefs: We...

Study smarter with the SolutionInn App



