Q1 Combustion of ethanol. The Lewis Structure of the molecule ethanol is presented in Figure Q1.1....
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
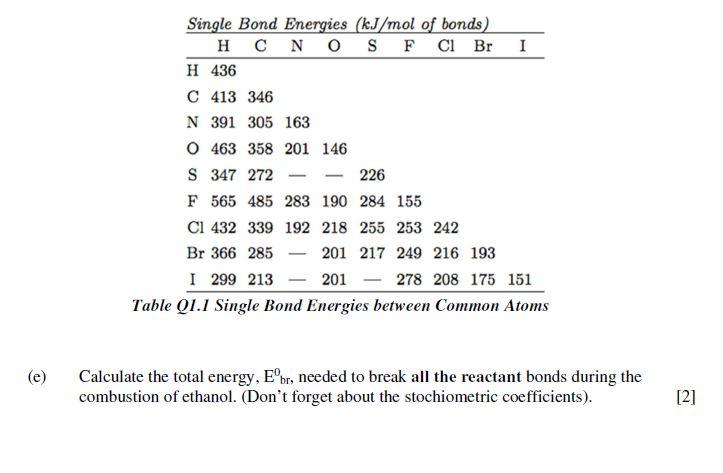
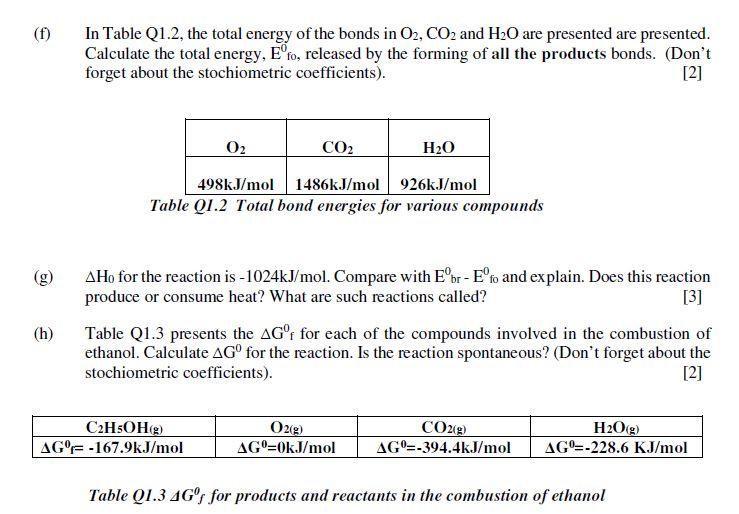
Q1 Combustion of ethanol. The "Lewis Structure" of the molecule ethanol is presented in Figure Q1.1. How many electron bonding pairs and electron lone pairs are present in the molecule? How many electrons is this in total? [2] (a) (b) (c) (d) H H H H-C-C H H Figure Q1.1 Lewis structure of Ethanol -H Carbon (C) has 4 electrons on its valence shell whilst hydrogen (H) has 1 and oxygen (0) has 6. How many valence electrons are there in ethanol? [2] The reaction of combustion of ethanol is presented in the reaction equation below. Balance the equation. [2] C₂H5OH(g) + O2(g) = CO2(g) + H₂O(g) In Table Q1.1, the energy for single bonds between common atoms are presented. Breaking bonds requires energy while forming bonds releases energy. Using Table 1, show that the energy required for the breaking of all the bonds in ethanol is equal to 3232kJ/mol. [5] (e) Single Bond Energies (kJ/mol of bonds) HCNOSF CI Br H 436 C 413 346 N 391 305 163 O 463 358 201 146 S 347 272 226 F 565 485 283 190 284 155 Cl 432 339 192 218 255 253 242 Br 366 285 201 217 249 216 193 I I 299 213 201 278 208 175 151 Table Q1.1 Single Bond Energies between Common Atoms Calculate the total energy, Eºbr, needed to break all the reactant bonds during the combustion of ethanol. (Don't forget about the stochiometric coefficients). [2] (f) (h) In Table Q1.2, the total energy of the bonds in O2, CO2 and H₂O are presented are presented. Calculate the total energy, Eºfo, released by the forming of all the products bonds. (Don't forget about the stochiometric coefficients). [2] CO₂ H₂O 498kJ/mol 1486kJ/mol 926kJ/mol Table Q1.2 Total bond energies for various compounds 0₂ Ho for the reaction is -1024kJ/mol. Compare with Eºbr - Eºfo and explain. Does this reaction produce or consume heat? What are such reactions called? [3] Table Q1.3 presents the AG°f for each of the compounds involved in the combustion of ethanol. Calculate AGO for the reaction. Is the reaction spontaneous? (Don't forget about the stochiometric coefficients). [2] C₂H5OH(g) AG% -167.9kJ/mol O2(g) AGO=0kJ/mol CO2(g) H₂O(g) AG=-394.4kJ/mol AG-228.6 KJ/mol Table Q1.3 4Gº for products and reactants in the combustion of ethanol (k) Calculate the standard change in entropy ASO for this reaction at 25°C. Explain why the entropy is positive. Q1 Combustion of ethanol. The "Lewis Structure" of the molecule ethanol is presented in Figure Q1.1. How many electron bonding pairs and electron lone pairs are present in the molecule? How many electrons is this in total? [2] (a) (b) (c) (d) H H H H-C-C H H Figure Q1.1 Lewis structure of Ethanol -H Carbon (C) has 4 electrons on its valence shell whilst hydrogen (H) has 1 and oxygen (0) has 6. How many valence electrons are there in ethanol? [2] The reaction of combustion of ethanol is presented in the reaction equation below. Balance the equation. [2] C₂H5OH(g) + O2(g) = CO2(g) + H₂O(g) In Table Q1.1, the energy for single bonds between common atoms are presented. Breaking bonds requires energy while forming bonds releases energy. Using Table 1, show that the energy required for the breaking of all the bonds in ethanol is equal to 3232kJ/mol. [5] (e) Single Bond Energies (kJ/mol of bonds) HCNOSF CI Br H 436 C 413 346 N 391 305 163 O 463 358 201 146 S 347 272 226 F 565 485 283 190 284 155 Cl 432 339 192 218 255 253 242 Br 366 285 201 217 249 216 193 I I 299 213 201 278 208 175 151 Table Q1.1 Single Bond Energies between Common Atoms Calculate the total energy, Eºbr, needed to break all the reactant bonds during the combustion of ethanol. (Don't forget about the stochiometric coefficients). [2] (f) (h) In Table Q1.2, the total energy of the bonds in O2, CO2 and H₂O are presented are presented. Calculate the total energy, Eºfo, released by the forming of all the products bonds. (Don't forget about the stochiometric coefficients). [2] CO₂ H₂O 498kJ/mol 1486kJ/mol 926kJ/mol Table Q1.2 Total bond energies for various compounds 0₂ Ho for the reaction is -1024kJ/mol. Compare with Eºbr - Eºfo and explain. Does this reaction produce or consume heat? What are such reactions called? [3] Table Q1.3 presents the AG°f for each of the compounds involved in the combustion of ethanol. Calculate AGO for the reaction. Is the reaction spontaneous? (Don't forget about the stochiometric coefficients). [2] C₂H5OH(g) AG% -167.9kJ/mol O2(g) AGO=0kJ/mol CO2(g) H₂O(g) AG=-394.4kJ/mol AG-228.6 KJ/mol Table Q1.3 4Gº for products and reactants in the combustion of ethanol (k) Calculate the standard change in entropy ASO for this reaction at 25°C. Explain why the entropy is positive.
Expert Answer:
Answer rating: 100% (QA)
1 Electronic configuration of various atom in ethanol molecule Carbon C 1s2 2s2 2p2 Hydrogen H 1s1 Oxygen O 1s2 2s2 2p4 Number of electrons in various atoms C 6 H 1 O 8 Number of valence electrons ele... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
please try to solve this question using Matlab and also explain it
-
Please write in complete sentences unless otherwise directed. 1. Benchmarking is an extremely useful business tool that helps organizations to stay competitive. Explain what benchmarking means. How...
-
PLEASE SOLVE ALL THE PARTS Q3). Given the function below complete the following tasks: y = f(x) = x + 4x? 20x 48 %3D Graph the function showing all intercepts, turning points, point(s) of...
-
What Codification citation indicates how unrealized gains or losses from changes in fair value affect net income when investments are classified as available-for-sale?
-
1. If the parties did not wish to allow e-mail to serve as an appropriate method to modify the contract, what working could have been used in Stevens employment agreement? 2. Could the principle that...
-
Constant Wall heat flux for turbulent flow between two parallel plates (a) Work through the development in S13.4, and then perform a similar derivation for turbulent flow in a thin slit shown in Fig....
-
Police in Albemarle County, Virginia, were on the lookout for a stolen orange and black motorcycle that had eluded them in two previous traffic incidents. Officer David Rhodes drove past the home of...
-
Gregs Bicycle Shop has the following transactions related to its top-selling Mongoose mountain bike for the month of March 2015: Required: 1. Calculate ending inventory and cost of goods sold at...
-
From the following ER diagram a) Translate it into a relational model b) Normalize it into BCNF (if possible) user id email name item_id description AuctionStartDate password phone_no age Browses 1...
-
It is the end of December and Natalie has been in touch with her grandmother. Her grandmother is curious to know if Natalie has been profitable and if Natalie requires another loan to help finance...
-
Fractal Design using Turtle Graphics: A four-step algorithm creates this fractal: Specify an intricacy level, a positive integer for the fractal. Start with a straight line. This line is called level...
-
Based on Apple vs Samsung lawsuit briefly explain the conflict scenario (facts of the case - background story) along with the parties involved in the negotiation process and then address the...
-
A dosimeter unit is tested for failure and median life of 6000 hours was found. Previous experience with these shows the time to failure is Weibull distributed with a shape factor of 3. What is the...
-
How does the design of facilities, including urban form, transportation, and parks influences physical activity?List some urban design strategies that can encourage more pedestrian and bicycle trips.
-
Transportation has an irrefutable importance to an economy. Explain transportation's role in an economy. Support your claim with evidence of transportation's impact on an economy (provide a positive...
-
The measured dimensions of a board are ( 1 7 . 6 pm 0 . 2 ) cm by ( 1 3 . 8 pm 0 . 1 ) cm . What is its area?
-
Wellington Jumbo Crab's stock currently sells for $70 per share with 1 million shares outstanding. The company wants to raise new equity using rights offer. To purchase a new share, a stockholder...
-
Pearson Education, a publisher of college textbooks, would like to know if students prefer traditional textbooks or digital textbooks. A random sample of students was asked their preference and the...
-
Cyclopropane and propylene are isomers (see Exercise 19.6) that both have the formula C3H6. Based on the molecular structures shown, which of these isomers would you expect to have the higher...
-
(a) What is electrolysis? (b) Are electrolysis reactions thermodynamically spontaneous? Explain. (c) What process occurs at the anode in the electrolysis of molten NaCl?
-
A deep-sea diver uses a gas cylinder with a volume of 10.0 L and a content of 51.2 g of O2 and 32.6 g of He. Calculate the partial pressure of each gas and the total pressure if the temperature of...
-
Smart Manufacturing Systems Pty Ltds accountant recently prepared the following data from the companys accounting records for the year ended 30 June 2019. Factory overhead is applied at the rate of...
-
During the year ended 30 June 2019, Beautiful Bottles Pty Ltd incurred the following costs in connection with its production activities. Required (a) Calculate the relationship between factory...
-
Telecommunications company Toronto Ltd signed a 15year deal to sell capacity on its cable network to a rival company for $200 million. The deal was completed on the last day of Toronto Ltds financial...

Study smarter with the SolutionInn App


