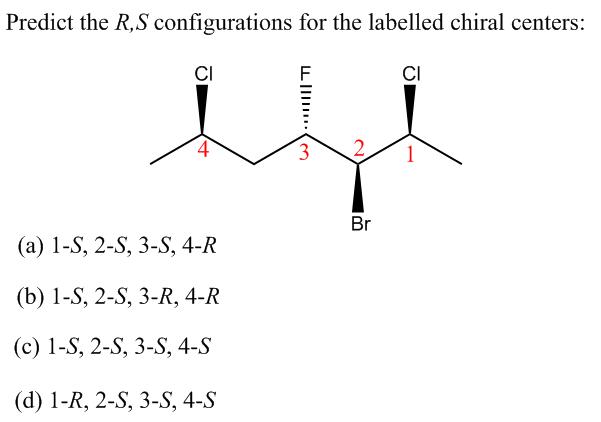
Predict the R,S configurations for the labelled chiral centers: CI CI 3. Br (a) 1-S, 2-S,...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
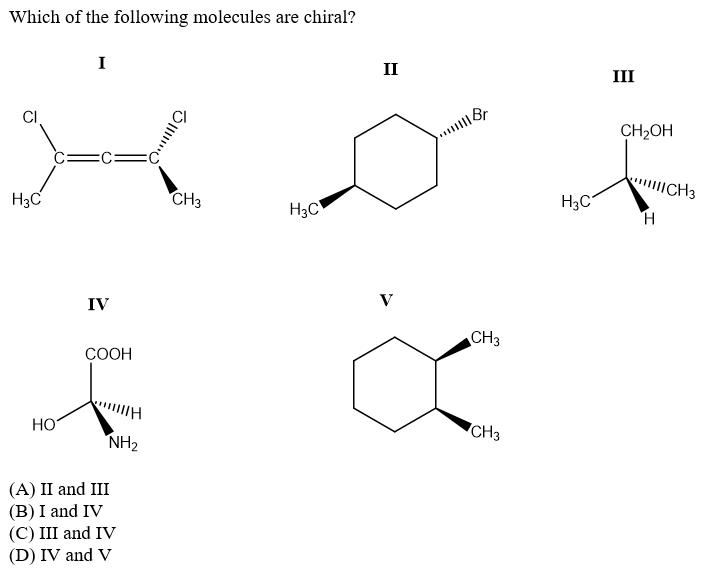
Predict the R,S configurations for the labelled chiral centers: CI CI 3. Br (a) 1-S, 2-S, 3-S, 4-R (b) 1-S, 2-S, 3-R, 4-R (c) 1-S, 2-S, 3-S, 4-S (d) 1-R, 2-S, 3-S, 4-S What is the eigen value of the function e* when operated on the operator d" / d" ? (A) a" (В) ха" (C) a'e* (D) a" / e* Which of the following molecules are chiral? I II III CH2OH CH3 H3C CH3 H3C H3C H. IV V CH3 COOH HO CH3 NH2 (A) II and III (B) I and IV (C) III and IV (D) IV and V Predict the R,S configurations for the labelled chiral centers: CI CI 3. Br (a) 1-S, 2-S, 3-S, 4-R (b) 1-S, 2-S, 3-R, 4-R (c) 1-S, 2-S, 3-S, 4-S (d) 1-R, 2-S, 3-S, 4-S What is the eigen value of the function e* when operated on the operator d" / d" ? (A) a" (В) ха" (C) a'e* (D) a" / e* Which of the following molecules are chiral? I II III CH2OH CH3 H3C CH3 H3C H3C H. IV V CH3 COOH HO CH3 NH2 (A) II and III (B) I and IV (C) III and IV (D) IV and V
Expert Answer:
Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
Which of the following molecules are chiral? I II III CH2OH CH3 H3C CH3 H3C H3C H. IV V CH3 COOH HO CH3 NH2 (A) II and III (B) I and IV (C) III and IV (D) IV and V
-
1. Which of the following molecules are chiral? a. 3-Bromopentane b. 1,3-Dibromopentane c. 3-Methylhex-1-ene d. cis-1,4-Dimethylcyclohexane e. 2,4-Dimethylheptane f. 5-Ethyl-3,3-dimethylheptane g....
-
What is the shear capacity of the RC beam described below considering the steel reinforcement and using the formula: VRsyAw 2fyd cot 8/s The shear reinforcement in the beam is provided by sets of...
-
A coil with 190 turns, a radius of 5.0 cm, and a resistance of 12 Ω surrounds a solenoid with 230 turns / cm and a radius of 4.5 cm (Fig. 21-65). The current in the solenoid changes at a...
-
Find the beginning of year NAV for AAGSX (Table 15 - 4).
-
Before beginning this assignment, read the GROW AI case carefully. Also, read the 2 HBR articles on Talent Analytics (Davenport et al. and the Contractor article). This is a TEAM assignment and must...
-
The Arcadia Company bid on a multiyear contract to construct the new hockey arena for the Phoenix Coyotes. The contract required that construction begin not later than January 1, 2015, and be...
-
You are to record transactions completed by Fabulous Furnishings during February of this year. Beginning balances for the accounts listed below have been provided in your Working Papers. This Company...
-
A study by the department of education of a certain state was trying to determine the mean SAT scores of the graduating high school seniors. The study examined the scores of a random sample of 84...
-
Eleven-Eleven, Inc. creates guided meditation programs for individuals. On November 30, 20Y9, the balances of selected accounts of Eleven-Eleven, Inc. are as follows: Prepare a classified balance...
-
Harry funds a charitable gift annuity with $100K paying his nephew $4K annually for the lifetime of niece Helen. Describe the gift tax consequences if any
-
A three-year 10 % annual coupon bond has a continuously compounded yield of 13%. (a) What is the bond price per $100 par? (b) What is its duration (D)? Explain the meaning of D. (c) Suppose the yield...
-
Interco is a publicly traded firm with two main operating divisions in paper products and biotechnology. The paper division generates EBIT of $12M per year and biotech generates EBIT of $3M....
-
1.Why is it easier for someone to perpetrate fraud using a journal entry than with a ledger? 2.Which enterprise resource planning (ERP) systems have you used? 3.What methods have you used for...
-
The Smartwatch Group predicted 250% annual growth in sales of the smartwatch industry for several years. The industry sales were low till middle of 2014 (about 700,000 smartwatches shipped...
-
(25 points) Estimate replacement cost new given the following information. Gross building area = 30,000 sqft Height = 3 stories Base Building Cost = $31/sqft Air filtration system = $15/sqft Height...
-
Gerald has taken out a loan of $100,000 today to start a business. He has agreed to repay the loan on the following terms: Repayments will be made on a monthly basis. The first repayment will be made...
-
Imagine you are the HR manager at a company, and a female employee came to you upset because she felt a male coworker was creating a hostile work environment by repeatedly asking her out on dates...
-
Give common or systematic names for each of the following compounds: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) CH3N+H3 CH3CO2 - (l) (m) (n) NH2 HIN NH2 CH3 NH2 OCH NIH N NH2 SO NH2 NH2 CH3
-
Outline a synthesis of propene from each of the following: (a) Propyl chloride (b) Isopropyl chloride (c) Propyl alcohol (d) Isopropyl alcohol (e) 1, 2-Dibromopropane (f) Propyne
-
Provide a structural formula for the product from each of the following reactions. (a) (b) (c) (d) (e) (f) (1) LDA (2) CH,CH Br. CH,COH NaH Cl Br2 (excess), NaOH (1) LDA (3) H20 ), NaoH
-
Objections to monopolies do not include which of the following? a. They reduce output below the efficient level of output that would be produced in perfect competition. b. They reduce the price below...
-
If regulators set a price according to marginal cost pricing, the firm will a. earn positive economic profits. b. make zero economic profits. c. suffer an economic loss. d. earn the same level of...
-
What is the objective of antitrust policy?

Study smarter with the SolutionInn App




