Consider the simple Linde-Hampson cycle shown in the figure. The gas is nitrogen and it is...
Fantastic news! We've Found the answer you've been seeking!
Question:
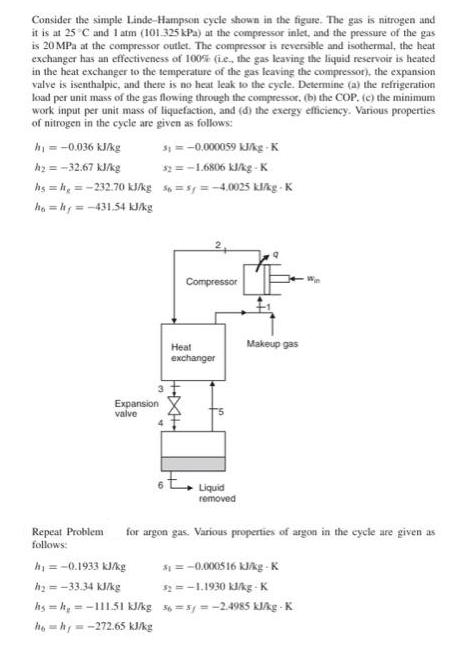
Transcribed Image Text:
Consider the simple Linde-Hampson cycle shown in the figure. The gas is nitrogen and it is at 25°C and 1 atm (101.325 kPa) at the compressor inlet, and the pressure of the gas is 20 MPa at the compressor outlet. The compressor is reversible and isothermal, the heat exchanger has an effectiveness of 100% (ie, the gas leaving the liquid reservoir is heated in the heat exchanger to the temperature of the gas leaving the compressor), the expansion valve is isenthalpic, and there is no heat leak to the cycle. Determine (a) the refrigeration load per unit mass of the gas flowing through the compressor, (b) the COP. (c) the minimum work input per unit mass of liquefaction, and (d) the exergy efficiency. Various properties of nitrogen in the cycle are given as follows: h₁ = -0.036 kJ/kg h₂ = -32.67 kJ/kg hs=h₂=-232.70 kJ/kg ho=hy=-431.54 kJ/kg Repeat Problem follows: Expansion valve 31 = -0,000059 kJ/kg-K 32 = -1.6806 kJ/kg-K h₁ = -0.1933 kJ/kg h₂=-33.34 kJ/kg =s=-4.0025 kl/kg-K Compressor Heat exchanger Makeup gas Liquid removed for argon gas. Various properties of argon in the cycle are given as =-0.000516 kJ/kg-K $2=-1.1930 kJ/kg-K hs=h₂=-111.51 kJ/kg 36=5y=-2.4985 kJ/kg-K ho=hy=-272.65 kJ/kg Consider the simple Linde-Hampson cycle shown in the figure. The gas is nitrogen and it is at 25°C and 1 atm (101.325 kPa) at the compressor inlet, and the pressure of the gas is 20 MPa at the compressor outlet. The compressor is reversible and isothermal, the heat exchanger has an effectiveness of 100% (ie, the gas leaving the liquid reservoir is heated in the heat exchanger to the temperature of the gas leaving the compressor), the expansion valve is isenthalpic, and there is no heat leak to the cycle. Determine (a) the refrigeration load per unit mass of the gas flowing through the compressor, (b) the COP. (c) the minimum work input per unit mass of liquefaction, and (d) the exergy efficiency. Various properties of nitrogen in the cycle are given as follows: h₁ = -0.036 kJ/kg h₂ = -32.67 kJ/kg hs=h₂=-232.70 kJ/kg ho=hy=-431.54 kJ/kg Repeat Problem follows: Expansion valve 31 = -0,000059 kJ/kg-K 32 = -1.6806 kJ/kg-K h₁ = -0.1933 kJ/kg h₂=-33.34 kJ/kg =s=-4.0025 kl/kg-K Compressor Heat exchanger Makeup gas Liquid removed for argon gas. Various properties of argon in the cycle are given as =-0.000516 kJ/kg-K $2=-1.1930 kJ/kg-K hs=h₂=-111.51 kJ/kg 36=5y=-2.4985 kJ/kg-K ho=hy=-272.65 kJ/kg
Expert Answer:

Related Book For 

South Western Federal Taxation 2015 Essentials of Taxation Individuals and Business Entities
ISBN: 9781285438290
18th edition
Authors: James Smith, William Raabe, David Maloney, James Young
Posted Date:
Students also viewed these accounting questions
-
In 2011, Charlie filed his 2010 state income tax return and paid taxes of $800. Also in 2011, Charlie's employer withheld state income tax of $750 from Charlie's salary. In 2012, Charlie filed his...
-
A question on a state income tax return asks the taxpayer if he or she made any out-of-state Internet or mail-order catalog purchases during the year. The question requires a yes or no answer, and if...
-
Prepare Lisa and Matt Davis Form 1040 for tax year 2020 based on the facts provided below. Please limit your analysis to the facts provided. Lisa (birthdate 3/15/1981) and Matt (birthdate 7/2/1982)...
-
A car of mass m starts from rest and accelerates so that the instantaneous power delivered to the car has a constant magnitude P. The instantaneous velocity of this car is proportional to : (a) t'P 0...
-
Independent random samples of n1 = n2 = n observations are to be selected from each of two binomial populations 1 and 2. If you wish to estimate the difference in the two population proportions...
-
The figure shows an element of soil at the interface between two dry sand layers on a 28 slope. The interface is 10 ft below the ground surface, and for both sand layers the friction angle is 34 and...
-
A tattoo shop has the following revenue stream over a 5 -year period: $\$ 9,000, \$ 17,000, \$ 19,000, \$ 11,000$, and $\$ 22,000$. What is the EUAW of this business (assume $\mathrm{i}=12 \%$ ).
-
In Mordica Company, total materials costs are $33,000, and total conversion costs are $54,000. Equivalent units of production are materials 10,000 and conversion costs 12,000. Compute the unit costs...
-
You have purchased one call and one put option on the same underlying stock. The strike price of the call option is 195 SEK and the strike price of the put option is 205 SEK. a)What is the minimum...
-
Three entrepreneurs were looking to start a new brewpub near Sacramento, California, called Roseville Brewing Company (RBC). Brewpubs provide two products to customersfood from the restaurant segment...
-
Dr. Marvin wrote a book called "Baby Steps" that was published by Touchy Feely Book Co. The parties met in New York to sign the contract. The contract stipulated the parties would use the law of New...
-
What are the benefits of reusable components?
-
Consider the buyer hot buttons . What are the implications for Betty Crocker? What new business areas might be considered given each hot button? Answer the same questions for a grocery store chain...
-
What is vendor-managed inventory?
-
Thirty samples of size 3, available in the worksheet C17 Excel P40 Data in MindTap were taken from a machining process over a 15-hour period. Construct control charts using the Excel template...
-
Hospital administrators wanted to understand and better control the waiting time of patients in the emergency department (ED). To do this, they constructed x -chart and R-charts by sampling the...
-
1 2 3 -2 1-5 -4 -5 Let A = -7 5r-19 z- 10 -7 11 -5r+27 -r+22 r+18 For which value of x is A invertible?
-
Prepare a stock card using the following information A company is registered for GST which it pays quarterly, assume GST was last paid on the 30th of June 2019. It uses weighted average cost...
-
Ida, who has AGI of $80,000 before considering rental activities, is active in three separate real estate rental activities. Ida has a marginal tax rate of 28%. She has $12,000 of losses from...
-
In 2014, Rose, Inc., has QPAI of $4 million and taxable income of $3 million. Rose pays independent contractors $500,000. Rose's W-2 wages are $600,000, but only $400,000 of the wages are paid to...
-
Clint, a self-employed engineering consultant, is contemplating purchasing an old building for renovation. After the work is completed, Clint plans to rent out two thirds of the floor space to...
-
According to the monetary approach to the balance of payments, what type of economic policies would help a country resolve a balance of trade deficit?
-
If two countries had identical term structures of interest rates, what is the expected future exchange rate change between the two currencies?
-
Assume that the cost of a particular basket of goods is equal to $108 in the United States and 14,000 in Japan. a. What should the U/$ exchange rate be according to absolute purchasing power parity?...

Study smarter with the SolutionInn App


