Question: Problem 1 Reversible process A reversible steady-state device receives a flow of 1.5kg/s of air at 400K, 450kPa, and the air leaves at 600K,
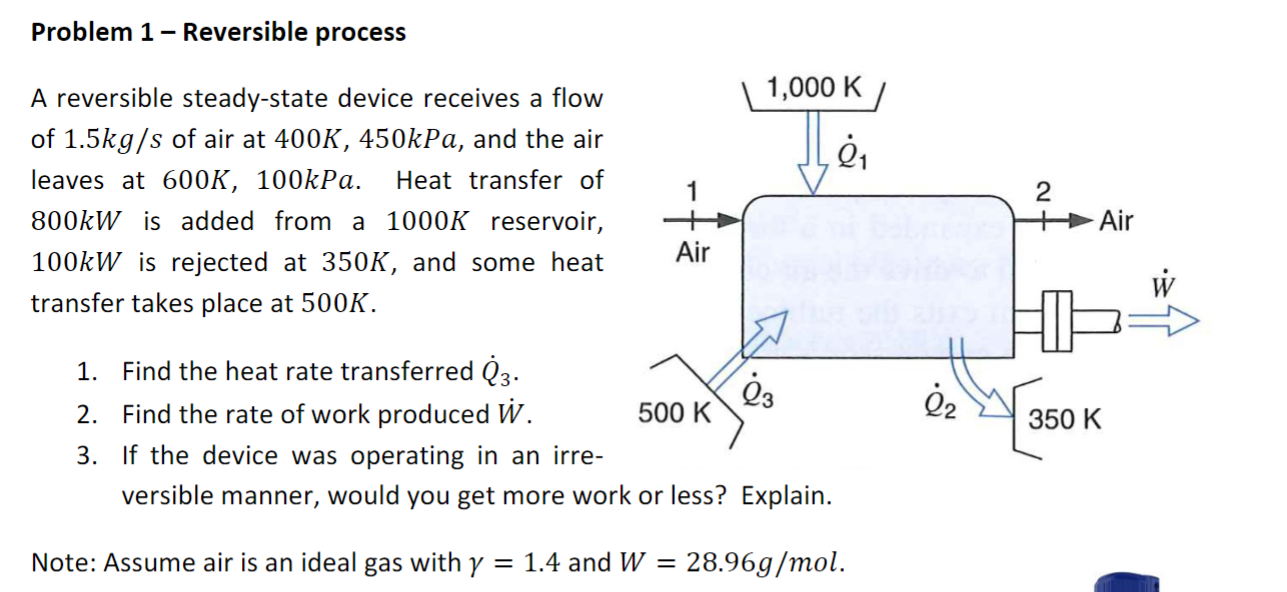
Problem 1 Reversible process A reversible steady-state device receives a flow of 1.5kg/s of air at 400K, 450kPa, and the air leaves at 600K, 100kPa. Heat transfer of 800kW is added from a 1000K reservoir, 100kW is rejected at 350K, and some heat transfer takes place at 500K. 1. Find the heat rate transferred Q3. Air 1,000 K/ 2. Find the rate of work produced W. 2 Air W 23 2 500 K 350 K 3. If the device was operating in an irre- versible manner, would you get more work or less? Explain. Note: Assume air is an ideal gas with y = 1.4 and W = 28.96g/mol.
Step by Step Solution
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


