Problem 12 A 6.90 M solution of KOH in water contains 30% by mass of KOH....
Fantastic news! We've Found the answer you've been seeking!
Question:
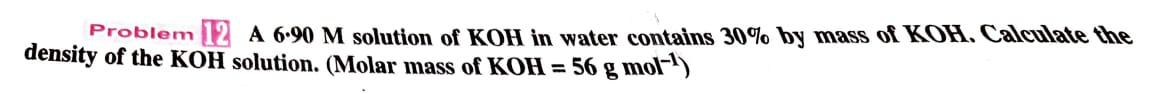
Transcribed Image Text:
Problem 12 A 6.90 M solution of KOH in water contains 30% by mass of KOH. Calculate the density of the KOH solution. (Molar mass of KOH= 56 g mol-¹) Problem 12 A 6.90 M solution of KOH in water contains 30% by mass of KOH. Calculate the density of the KOH solution. (Molar mass of KOH= 56 g mol-¹)
Expert Answer:
Answer rating: 100% (QA)
density can be calculated by taking the ratio of Mass and density When one mole of solute is dissolv... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Calculate the number-average molar mass and the mass-average molar mass of a mixture of two polymers, one having M = 62 kg mol-1 and the other M = 78 kg mol-1, with their amounts (numbers of moles)...
-
A certain tank contains water whose mass density is 1.94 slug/ft3. The tank's circular bottom area is A = 100 ft2. It is drained by an orifice in the bottom. The effective cross-sectional area of the...
-
A liquid solution contains 1 mol of CaCl2 and 25 mol of water. Using data from Pb. 12.37, determine the heat effect when an additional 1 mol of CaCl2 is dissolved isothermally in this solution. In...
-
Suppose we want both the sensitivity and specificity to be at least 70%. Use the ROC curve to identify the possible value(s) to use as the cutoff for identifying people with dementia, based on these...
-
Utilizing your critical thinking skills, solve the following problem: Three auditors checked into a hotel under one reservation. After paying $100 each to the hotel manager, they went to their...
-
The following production information is available for Porter Corporation: Budgeted production . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 150,000 units Actual...
-
Why does collusion pose unique prevention and detection challenges?
-
Copy Service Co. purchased a new color copier at the beginning of 2010 for $42,000. The copier is expected to have a five-year useful life and a $6,000 salvage value . The expected copy production...
-
Supposons un consommateur ayant une richesse W qui est distribue selon une loi de densit de probabilit fw (w). Montrez qu'on peut obtenir une approximation du cot du risque, CR, en utilisant: 1 CRrr...
-
The following state table is implemented using a ROM and two D flip-flops (falling edge triggered): (a) Draw the block diagram. (b) Write Verilog code that describes the system. Assume that the ROM...
-
Leonard Company is preparing its third quarter production budget. Projected sales in units are 600 for July, 680 for August, and 750 for September. Leonard's policy is to have finished goods...
-
Explain how an effective performance management system along with compensation and benefits can attract, develop and retain talented employees. Discuss labor and employment laws and regulations that...
-
You have been asked to send an email to Stacey Mintern. The email contained some background notes attached about an important meeting she has scheduled for tomorrow with the other departments. You...
-
A firm that is currently locating facilities in a large number of other countries in order to capitalize on lower production and distribution costs is at what level of global participation?...
-
Describe the legal, social, and psychological employment contracts in organisations. Citing examples, explain why it is important for HRM to manage each of these three contracts at the workplace....
-
What special compensation and benefits issues arise in international HRM ?
-
Niharika has been a salesperson for Rebecca Products since January 2005. She is paid solely by 10% commission. Her four sales were made in the last 52 weeks were: Sales Qrt 1 = $250,000 Sales Qrt 2 =...
-
Answer the following two independent questions. a. MM Corporation is considering several proposed investments for the coming budget year. MM produces electrical apparatus for industrial complexes....
-
The following plot shows the pH curves for the titrations of various acids with 0.10 M NaOH (all of the acids were 50.0-mL samples of 0.10 M concentration). a. Which pH curve corresponds to the...
-
Naturally occurring uranium is composed mostly of 238U and 235U, with relative abundances of 99.28% and 0.72%, respectively. The half-life for 238U is 4.5 X 109 years, and the half-life for is 7.1 X...
-
The common names and formulas for several substances are given below. What are the systematic names for these substances? a. Sugar of lead Pb(C2H3O2)2 b. Blue vitriol CuSO4 c. Quicklime CaO d. Epsom...
-
Cinnamon, Inc. recorded a total deferred tax asset in Year 3 of \($12\),301, off set by a \($12\),301 valuation allowance. Cinnamon most likely: A . fully utilized the deferred tax asset in Year 3. B...
-
Midland Brands issues three-year bonds dated January 1, 2015 with a face value of $5,000,000. Th e market interest rate on bonds of comparable risk and term is 3%. If the bonds pay 2.5% annually on...
-
A company issues $1,000,000 face value of 10-year bonds on January 1, 2015 when the market interest rate on bonds of comparable risk and terms is 5%. Th e bonds pay 6% interest annually on December...

Study smarter with the SolutionInn App


