Prove that the specific volume of the moisture air (v) can be estimated from the following...
Fantastic news! We've Found the answer you've been seeking!
Question:
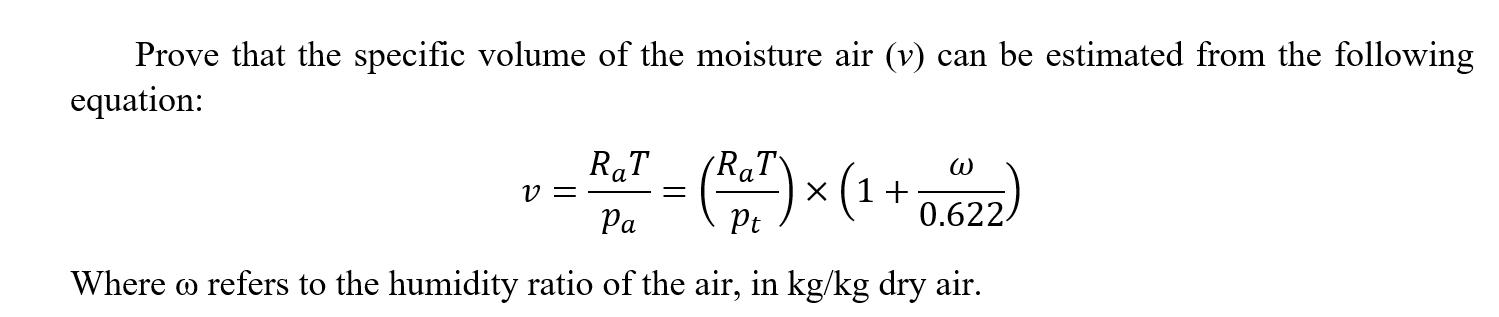
Transcribed Image Text:
Prove that the specific volume of the moisture air (v) can be estimated from the following equation: RaT - = (RaT) x (1 + Pa Pt Where o refers to the humidity ratio of the air, in kg/kg dry air. v= W 0.622/ Prove that the specific volume of the moisture air (v) can be estimated from the following equation: RaT - = (RaT) x (1 + Pa Pt Where o refers to the humidity ratio of the air, in kg/kg dry air. v= W 0.622/
Expert Answer:
Answer rating: 100% (QA)
SOLUTION To prove the equation v Ra T pa Ra T pt1 omega0622 where v represents the specific volume o... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder, ? Ronald W. Rousseau, ? Lisa G. Bullard
Posted Date:
Students also viewed these marketing questions
-
4. Crazy Soccer is the CFO of Bayern Gas, a large German manufacturer of industrial, commercial, and consumer gas products. Bayern Gas is privately owned, and its shares are not listed on an...
-
The conversion of the kinetic energy of wind to electricity may be an attractive alternative to the use of fossil fuels. Typically, wind causes the rotor of a turbine to turn, and a generator...
-
The air in a room is at 1 atm, 32C, and 60 percent relative humidity. Determine (a) The specific humidity, (b) The enthalpy (in kJ/kg dry air), (c) The wet-bulb temperature, (d) The dew-point...
-
Stems of the first land plants functioned primarily as transport and support structures. Modern stems have these functions also, plus several more. Describe the additional functions.
-
An analyst stated that, all else equal, increasing sample size will decrease both the standard error and the width of the confidence interval. The analyst's statement is correct in regard to: A. Both...
-
You have been asked by a client to review the records of Roberts Company, a small manufacturer of precision tools and machines. Your client is interested in buying the business, and arrangements have...
-
Steam at \(400^{\circ} \mathrm{F}, 70 \mathrm{psia}\), and \(100 \mathrm{lb} / \mathrm{hr}\) is compressed to 200 psia. The electrical work is \(4.1 \mathrm{~kW}\). Determine the: (a) Lost work (b)...
-
The Tiny Treasures Company must decide whether to continue selling a line of childrens shoes manufactured on a machine that has no other purpose. The machine has a current book value of $12,000, and...
-
S Each of the four independent situations below describes a sales-type lease in which annual lease payments of $100,000 are payable at the beginning of each year. Each is a finance lease for the...
-
You, CPA, work as an associate with Campbell and Associates LLP, a financial and business advisory firm. The board of directors of Pembroke Pulp and Paper Inc. (PPPI) has engaged your firm once again...
-
1. Identify three restrictions imposed on Indigenous people by the Indian Act. What was the impact of this Act on Indigenous Women and Children? 2. Describe how Indigenous peoples and Women advocated...
-
Now that you have completed the Interpersonal Skills Project this semester, let's take some time to reflect on all that you have accomplished. Consider your experience with the term-long skills...
-
Who is in your mind, a speaker in any time of history, who you liked in terms of their speech and delivery, and explain why you thought they were effective, inspirational, etc.
-
Find an example of data/claims related to your interests or your work. https://www.wral.com/survey-finds-7-of-north-carolina-teachers-plan-to-quit-after-this-school-year/20319953/ required Summarize...
-
A tech startup is entering the competitive field of AI-driven analytics. How would the founders articulate their mission and vision in the business plan to differentiate themselves from established...
-
Swifty Corporation borrowed $660000 from Liber Bank on January 1, 2019 in order to expand its mining capabilities. The five-year note required annual payments of $178200 and carried an annual...
-
2) Draw a diagram of a simple food web with three trophic levels and describe the difference food chain and a food web. between
-
The nitrogen atoms in N2 participate in multiple bonding, whereas those in hydrazine, N2H4, do not. (a) Draw Lewis structures for both molecules. (b) What is the hybridization of the nitrogen atoms...
-
Two systems contain water, acetone, and methyl isobutyl ketone in equilibrium at 25C. The first system contains equal masses of the three species, and the second one contains 9.0% acetone, 21.0%...
-
Benzaldehyde is produced from toluene in the catalytic reaction Dry air and toluene vapor are mixed and fed to the reactor at 350F and 1 atm. Air is supplied in 100% excess. Of the toluene fed to the...
-
A fuel gas containing 85.0 mole% methane and the balance ethane is burned completely with pure oxygen at 25C and the products are brought back down to 25C. (a) Suppose the reactor is continuous. Take...
-
Superheated steam available at \(800 \mathrm{psia}\) and \(600^{\circ} \mathrm{F}\) is to be expanded to a pressure of \(150 \mathrm{psia}\) at the rate of \(100,000 \mathrm{lb} / \mathrm{hr}\)....
-
Use a simulator to design a compression system with intercoolers to compress \(600 \mathrm{lb} / \mathrm{hr}\) of a mixture of \(95 \mathrm{~mol} \%\) hydrogen and \(5 \mathrm{~mol} \%\) methane at...
-
A certain bon vivant, wishing to ascertain the cause of his frequent hangovers, conducted the following experiment. On the first night, he drank nothing but whiskey and water; on the second night, he...

Study smarter with the SolutionInn App


