Pure water is used to dilute 152 lbm of a sulfuric acid stream that has an...
Fantastic news! We've Found the answer you've been seeking!
Question:
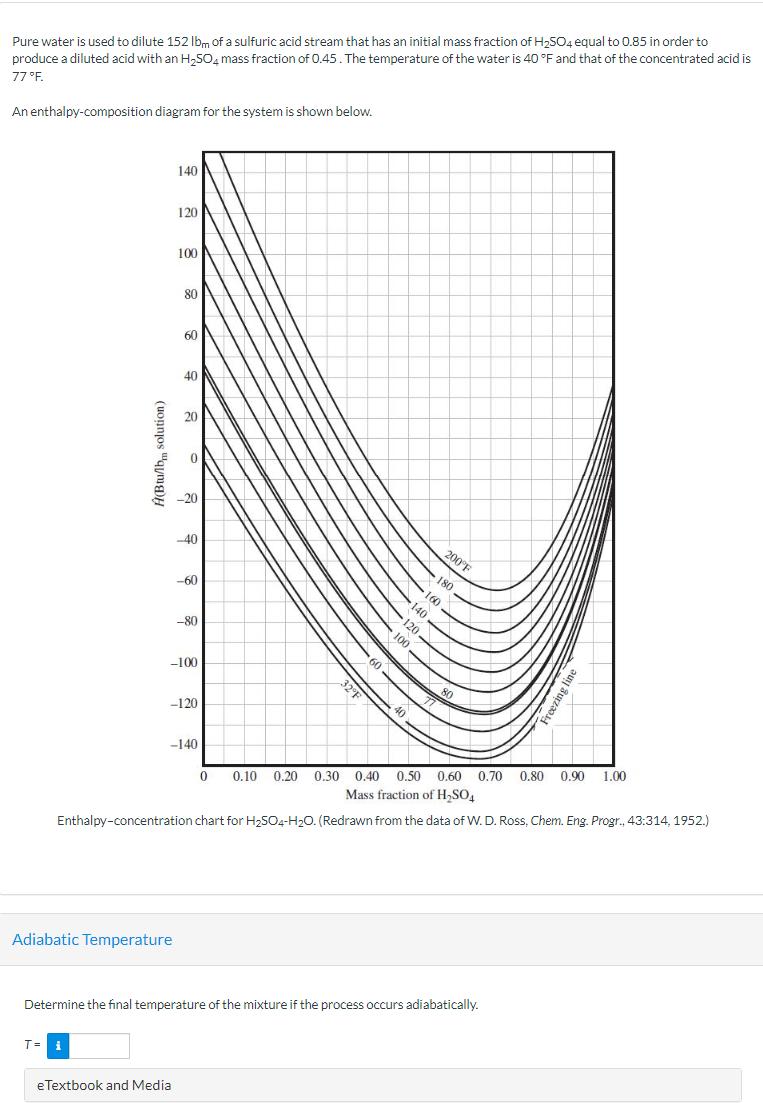
Transcribed Image Text:
Pure water is used to dilute 152 lbm of a sulfuric acid stream that has an initial mass fraction of H2SO4 equal to 0.85 in order to produce a diluted acid with an H₂504 mass fraction of 0.45. The temperature of the water is 40 °F and that of the concentrated acid is 77°F. An enthalpy-composition diagram for the system is shown below. Ĥ(Btu/lb solution) 140 120 100 80 60 40 20 -20 -40 -60 -80 -100 -120 -140 60 40 32°F 200°F 80 Freezing line 0 0.10 0.20 0.30 0.40 0.50 0.60 0.70 0.80 0.90 1.00 Mass fraction of H₂SO4 Enthalpy-concentration chart for H2504-H2O. (Redrawn from the data of W. D. Ross, Chem. Eng. Progr., 43:314, 1952.) Adiabatic Temperature Determine the final temperature of the mixture if the process occurs adiabatically. T= i eTextbook and Media Pure water is used to dilute 152 lbm of a sulfuric acid stream that has an initial mass fraction of H2SO4 equal to 0.85 in order to produce a diluted acid with an H₂504 mass fraction of 0.45. The temperature of the water is 40 °F and that of the concentrated acid is 77°F. An enthalpy-composition diagram for the system is shown below. Ĥ(Btu/lb solution) 140 120 100 80 60 40 20 -20 -40 -60 -80 -100 -120 -140 60 40 32°F 200°F 80 Freezing line 0 0.10 0.20 0.30 0.40 0.50 0.60 0.70 0.80 0.90 1.00 Mass fraction of H₂SO4 Enthalpy-concentration chart for H2504-H2O. (Redrawn from the data of W. D. Ross, Chem. Eng. Progr., 43:314, 1952.) Adiabatic Temperature Determine the final temperature of the mixture if the process occurs adiabatically. T= i eTextbook and Media
Expert Answer:
Answer rating: 100% (QA)
Determining the Final Temperature of the Mixture Heres how to determine the final temperature of the mixture in this adiabatic mixing process Calculate the total mass of the mixture Mass of concentrat... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
For your cabin in the wilderness, you decide to build a primitive refrigerator out of Styrofoam, planning to keep the interior cool with a block of ice that has an initial mass of24.0 kg. The box has...
-
Consider : You have been asked to evaluate whether your organization's current pay structure makes sense in view of what competing - address the following: How would you determine what organizations...
-
Offer some suggestions on where to get money to invest.
-
A system is shown in Figure E5.18(a). The response to a unit step, when K = 1, is shown in Figure E5.18(b). Determine the value of K so that the steady-state error is equal to zero. GIS) fs) ir 1.0 0...
-
Fun-Tastic Shows, Inc., is a company that hosts carnivals and similar events. Susan Swartwood, Crystal Groth, and a minor (named in the case as M.G.S.) attended Fun-Tastics Rhododendron Festival in...
-
Given the following project to landscape a new building site, (a) Draw a Gantt chart using MSP. (b) Find the critical path and project duration in days. (c) Given that each resource is assigned 100%...
-
Nick went out for dinner with a few friends. The restaurant bill should have been $175, but instead, the waitress brings the bill for $160. Find the absolute error and relative percentage error for...
-
- Assume that this assignment involves an existing business that was purchased by the new owner on 1 June 2019. - Note the list of transactions is in date order, left to right, line by line. - Use...
-
Listen to the two podcasts you were assignment please listen to: Episode Five: Housing Episode Three: Migration and Arrival aim to summarize your learning. What details stood out to you? What struck...
-
8. Your investment broker has offered you the opportunity to receive $1000 each year for 5 years. Suppose your required rate of return is 6%. What is the most you should be willing to pay today for...
-
Suppose one pound is trading for 2 . 0 0 euro in the spot market, the interest rate on a one - year deposit denominated in pounds is 2 percent per annum, and the interest rate for a comparable ( one...
-
15 postcards and 10 envelopes cost 1 dollar and 70 cents. An envelope is 2 cents more expensive than a postcard. What is the price of the postcard and the envelope?
-
Consider the following. f(x)=5x-5 Compare the values of Ay and dy if x changes from 1 to 1.05. (Round your answers to four decimal places.) Ay - dy = 42.1531 What if x changes from 1 to 1.02? (Round...
-
In a container that contains a fluid, and 2 thermometers are introduced, one in degrees Centigrade and another in degrees Fahrenheit, and both indicate the same reading. And its pressure is 1.0 bar...
-
The packaging division of a company having considered several alternative package designs for the company's new product has finally brought down their choices to two designs of which only one has to...
-
How do you think the rate of reaction, r A , will depend on species concentrations and on temperature?
-
View two of the safety acronym links and write an evaluation as to whether or not the link was useful. A graph of F subscript A 0 over negative r subscript A is shown. Initially, the value of F...
-
Revisit Problem P16-3 where the RTD function is a hemicircle. The liquid-phase reaction is first order with k1 = 0.8 min 1 . What is the conversion predicted by a. The tanks-in-series model? b. The...
-
A spring pendulum features a pendulum bob of mass \(m\) attached to one end of a spring of force-constant \(k\) and unstretched length \(R\). The other end of the spring is attached to a fixed point...
-
A pendulum is constructed from a bob of mass \(m\) on one end of a light string of length \(D\). The other end of string is attached to the top of a circular cylinder of radius \(R\) \((R <2 D /...
-
A plane pendulum is made with a plumb bob of mass \(m\) hanging on a Hooke'slaw spring of negligible mass, force constant \(k\), and unstretched length \(\ell_{0}\). The spring can stretch but is not...

Study smarter with the SolutionInn App


