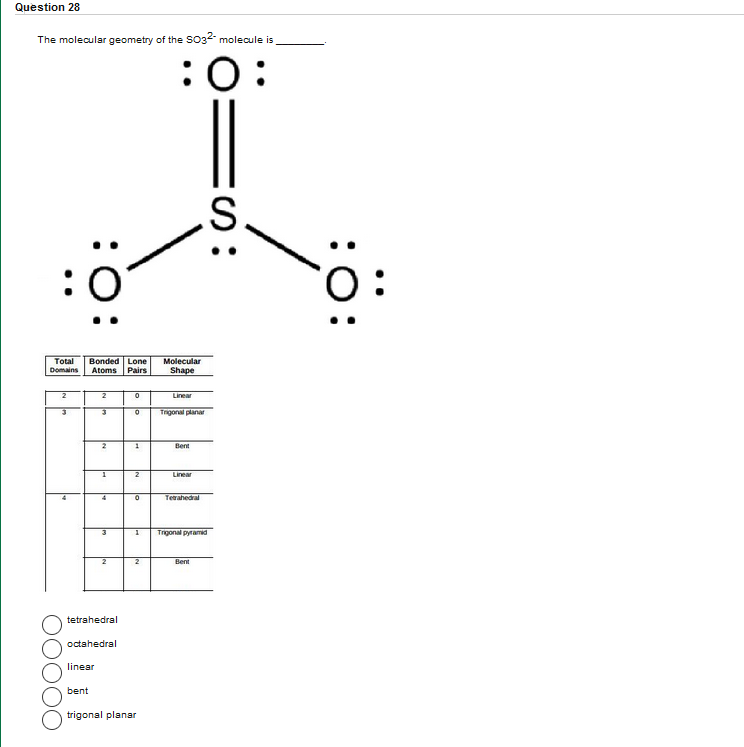
Question 28 The molecular geometry of the SO32- molecule is :O: :0 Total Domains 3 :O:...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Question 28 The molecular geometry of the SO32- molecule is :O: :0 Total Domains 3 :O: Bonded Lone Atoms Pairs bent 2 3 linear 2 1 3 tetrahedral 2 octahedral 0 0 1 2 0 1 2 trigonal planar Molecular Shape Linear Trigonalplanar Bent Linear Tetrahedral Trigonal pyramid Bent :S: S. :O: 0: Question 28 The molecular geometry of the SO32- molecule is :O: :0 Total Domains 3 :O: Bonded Lone Atoms Pairs bent 2 3 linear 2 1 3 tetrahedral 2 octahedral 0 0 1 2 0 1 2 trigonal planar Molecular Shape Linear Trigonalplanar Bent Linear Tetrahedral Trigonal pyramid Bent :S: S. :O: 0:
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Identify the electron group geometry and the molecular geometry of each molecule. a. H2S b. POCl3
-
Identify the electron group geometry and the molecular geometry of each molecule. a. CS2 b. H2S
-
Identify the electron group geometry and the molecular geometry of each molecule. a. HCN b. CCl4
-
Factor completely. p(p + 2) + p(p + 2) - 6(p + 2)
-
Refer to the information given in the preceding problem for Bodacious Bagels, Inc. Required: Redesign the bagel production process after eliminating the non-value-added costs. What new equipment...
-
What has been the financial incentive for the high-tech companies to agree not to recruit from one another? If the arrangement had not been challenges in court, would you consider them ethical? Why...
-
People immersed in strong unchanging magnetic fields occasionally report sensing a metallic taste. Some investigators suspect that motion in the constant field could produce a changing flux and a...
-
Suppose demand and supply are given by Qd = 50 P and Qs = P 10. a. What are the equilibrium quantity and price in this market? b. Determine the quantity demanded, the quantity supplied, and the...
-
MR Green is one of the resident for who you take care in a nursing home .He is recovering from a stroke and has a pressure ulcer . In addition he is disoriented and confuse . However he can feed...
-
Pathfinder College is a small liberal arts college that wants to improve its admissions process. In particular, too many of its incoming freshmen have failed to graduate for a variety of reasons,...
-
Al's Sport Store has sales of $897,400 and accounts receivable of $74,100. How many days, on average, does it take the firm to collect its receivables? (Round to the closest # of days)
-
Question 6 Determine the oxidation number of manganese (Mn) in each of the following compounds. [Select] [Select] +3 -3 +2 (1) Mn03 (2) MnO (3) KMnO4 (3) KMnO [Select] [Select] [Select c Question 7...
-
Question 1c List 20 different natural materials / resources that are familiar, that you would provide within your learning environment. Familiar natural materials / resources Satisfactory Not...
-
9. Assume a direct mapped cache, with four lines and the tag values for each lines are as below: Line 00 01 10 11 Tag 00110 00001 00000 Invalid The tag, line, and word field contains: 5, 2, and 5...
-
A horizontal lever of length 2 . 2 m is pivotedat its mid - point. An downward force of 1 3 N is acting at the right end of the lever. Calculate the torque acting on the lever. -
-
What is the minimum energy needed to change the speed of a 1600-kg sport utility vehicle from 15.0 m/s to 40.0 m/s?
-
1.00 kg of liquid water at 20.0 C is converted to superheated steam at 1200 C in a boiler operating at a constant pressure of 5.00 MPa. Using the temperature dependent heat capacity formula, how much...
-
Assume that a trial balance is prepared with an account balance of $21,360 listed as $21,630 and an account balance of $1,500 listed as $15,000. Identify the transposition and the slide.
-
Would the trial wave function have been a suitable choice for the calculations carried out in Section 21.4? Justify your answer. 3) 48) () %3D 0 < x < a
-
Explain why the spectator species M in Equation (25.5) is needed to make the reaction proceed.
-
Predict the number of chemically shifted 1 H peaks and the multiplet splitting of each peak that you would observe for nitromethane. Justify your answer.
-
Lewis Edwards decides to branch out on his own and set up his own private practice as an accountant. Events occurring in March 2019 are as follows. Ignore GST. Required (a) After analyzing the events...
-
On 1 March 2017, James Taylor decided to open Taylors Tailormade that makes suits, trousers and jackets, and repairs and alters clothes. He contributed for this purpose sewing equipment $46 000 and a...
-
Peters Personal Training Service had been in business for several years. In June 2019, as a result of a dispute with the owner, the accountant of the business disappeared and took all the records...

Study smarter with the SolutionInn App



