Question: Question 3 The normal boiling point for the substance in the phase diagram below is approximately Pressure (atm) O 0C 65C 35C 1- O
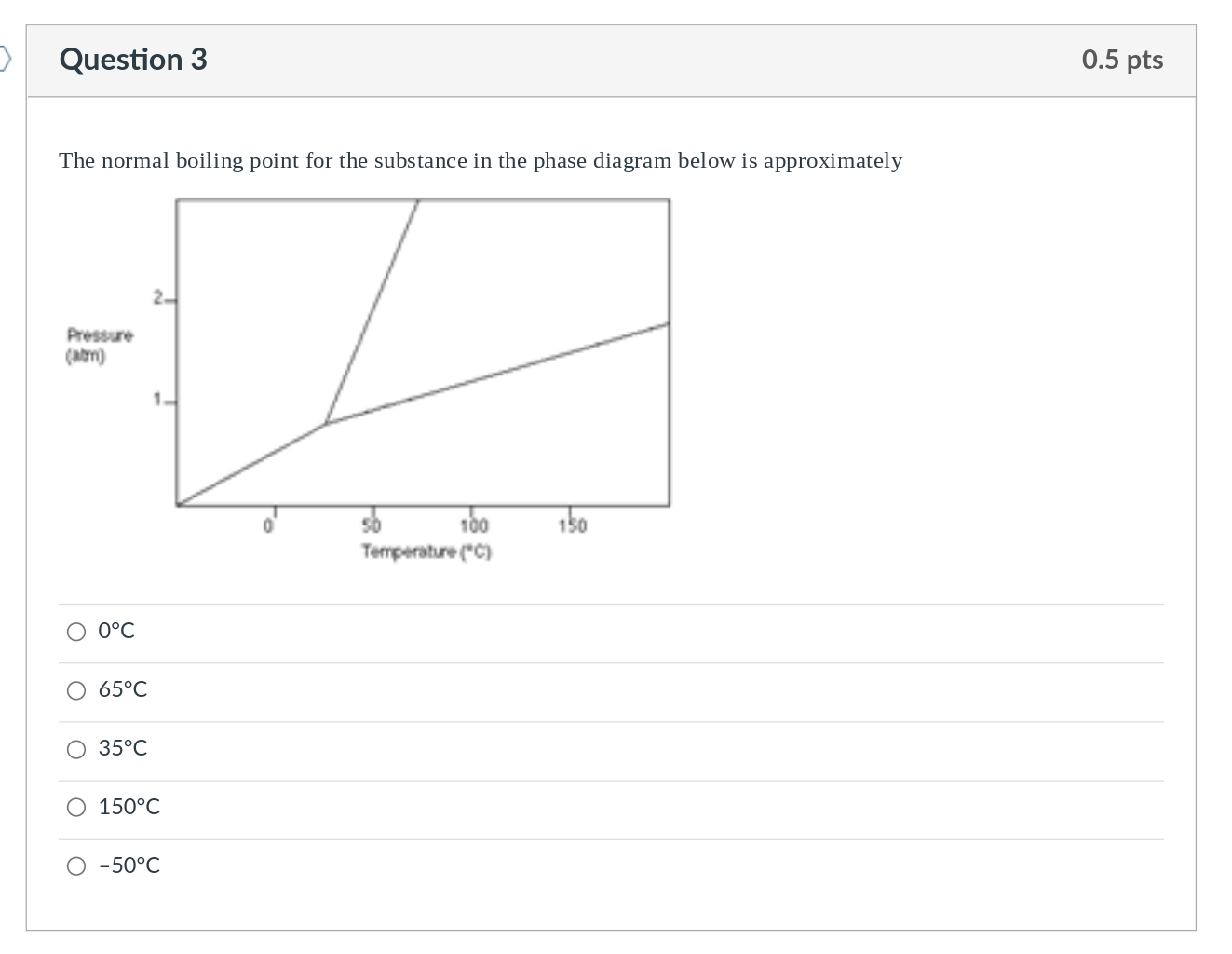
Question 3 The normal boiling point for the substance in the phase diagram below is approximately Pressure (atm) O 0C 65C 35C 1- O 150C O -50C 50 100 Temperature (C) 150 0.5 pts
Step by Step Solution
3.51 Rating (151 Votes )
There are 3 Steps involved in it
Answer Solution A phase transition involving liquid to gas change at 1 atm pressure is the no... View full answer

Get step-by-step solutions from verified subject matter experts


