Required information [The following information applies to the questions displayed below.] Daley Company prepared the following...
Fantastic news! We've Found the answer you've been seeking!
Question:
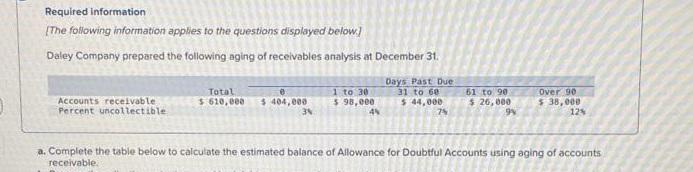
Transcribed Image Text:
Required information [The following information applies to the questions displayed below.] Daley Company prepared the following aging of receivables analysis at December 31. Days Past Due Total e Accounts receivable Percent uncollectible $610,000 $ 404,000 1 to 30 $ 98,000 31 to 60 $44,000 61 to 90 $ 26,000 Over 90 $ 38,000 3% 4% 7% 9% 12% a. Complete the table below to calculate the estimated balance of Allowance for Doubtful Accounts using aging of accounts receivable. Required information [The following information applies to the questions displayed below.] Daley Company prepared the following aging of receivables analysis at December 31. Days Past Due Total e Accounts receivable Percent uncollectible $610,000 $ 404,000 1 to 30 $ 98,000 31 to 60 $44,000 61 to 90 $ 26,000 Over 90 $ 38,000 3% 4% 7% 9% 12% a. Complete the table below to calculate the estimated balance of Allowance for Doubtful Accounts using aging of accounts receivable.
Expert Answer:

Posted Date:
Students also viewed these accounting questions
-
For rigid-body motion, the strains will vanish. Under these conditions, integrate the straindisplacement relations (7.6.1) to show that the most general form of a rigid-body motion displacement field...
-
Why should an organization select multiple metrics to measure performance? Why is there a need to measure performance using both short-run and long-run time horizons?
-
Below are the restated amounts of net income and retained earnings for Volunteers Inc. and Raiders Inc. for the period 20152024. Volunteers began operations in 2016, while Raiders began several years...
-
The stockholders' equity of Ranger Corporation at January 1 appears below: During the year, the following transactions occurred: May 12 Declared a 15 percent stock dividend; market value of the...
-
The treasury stock purchased in question 13 is resold by Chen, Inc. for $15,000. What effect does this transaction have on (a) Net income, (b) Total assets, (c) Total paid-in capital, and (d) Total...
-
Brooklyn has been contributing to a traditional IRA for seven years ( all deductible contributions ) and has a total of $ 3 0 , 0 0 0 in the account. In 2 0 2 3 , she is 3 9 years old and has decided...
-
Drew owns and operates an onion packing plant. To help reduce costs on labor and to increase efficiency, Drew is considering purchasing an automatic 50 lb. bagging machine and a machine that...
-
For each pair of the following second order system specifications obtain the location of the pair of poles in s - plane (a) % OS = 12%. T = 0.6sec. (b) % OS = 10%. Ip = 5sec. (c) T = 7%. T = 3sec. P
-
What is strategy execution? Why is good strategy execution so important? As a part of good strategy execution, why is it important to match the company's strategy and structure? What are some ways to...
-
How do Samsung take advantage of the various aspects of Integrated Marketing? What tools, print media, TV, social networks, do they use??
-
Write a two-page paper on how you would conduct a half-an-hour physical activity work-out session, indicating the specific target group you are working with. In this you must clearly explain all...
-
Analyze how Eataly's strategic planning contributed to the competitive advantage it has created, using the balance scorecard, SWOT, or PESTLE analysis, and the value chain to analyze its strengths or...
-
Explain how the following would impact DNA replication in prokaryotes and eukaryotes. 6.1 The absence of UTP. 6.2 Mutation in the origin of replication sequence.
-
Chao, Louis, and Mari, unrelated individuals, own all of the shares of Cerise Corporation. All three shareholders have been active in the management of Cerise since its inception. In the current...
-
Repeat Example 15-10 but with a mass transfer coefficient that is 10 times larger (use \(\delta=0.001 \mathrm{~m})\). Report \(\mathrm{x}_{\mathrm{NH} 3}, \mathrm{y}_{\mathrm{NH} 3, \text { surface...
-
A particle of pure \(\mathrm{NaCl}\) is dissolving in an aqueous liquid solution at \(18^{\circ} \mathrm{C}\). The dissolution of the particle is controlled by mass transfer. The system is vigorously...
-
Calculate the value of Maxwell-Stefan diffusivity for ethanol water at \(40^{\circ} \mathrm{C}\) for ethanol mole fractions of \(0.0,0.2,0.3,0.4,0.7\), and 1.0 . The Fickian diffusivities are...

Study smarter with the SolutionInn App

