Question: Using Figure 1 below and solve the following questions: 1.1. Which salt display discontinuities in their solubility curves? Explain what contributes to the occurrence
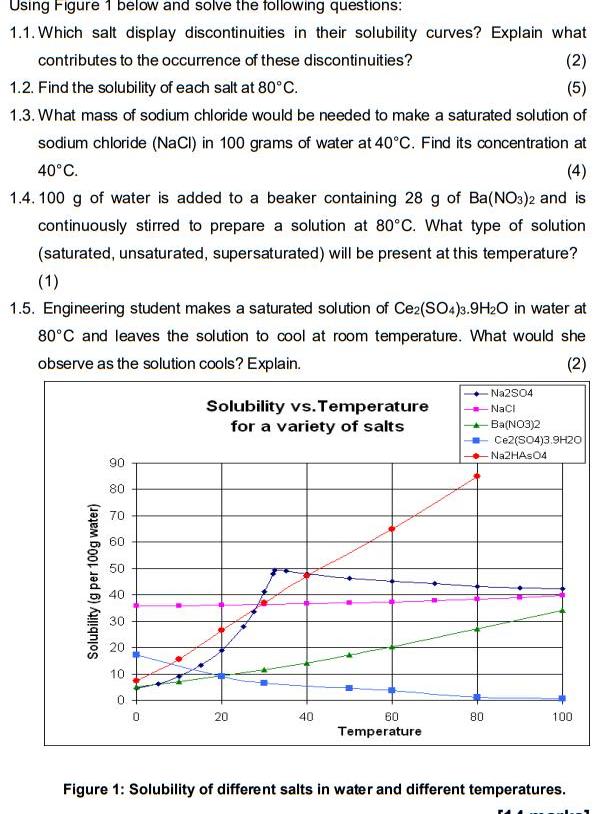
Using Figure 1 below and solve the following questions: 1.1. Which salt display discontinuities in their solubility curves? Explain what contributes to the occurrence of these discontinuities? (2) 1.2. Find the solubility of each salt at 80c. (5) 1.3. What mass of sodium chloride would be needed to make a saturated solution of sodium chloride (NaCI) in 100 grams of water at 40C. Find its concentration at 40C. (4) 1.4. 100 g of water is added to a beaker containing 28 g of Ba(NO3)2 and is continuously stirred to prepare a solution at 80C. What type of solution (saturated, unsaturated, supersaturated) will be present at this temperature? (1) 1.5. Engineering student makes a saturated solution of Ce2(SO4)3.9H2O in water at 80C and leaves the solution to cool at room temperature. What would she observe as the solution cools? Explain. (2) Na2S04 Solubility vs. Temperature for a variety of salts Naci - Ba(NO3)2 Ce2(S04)3.9H20 Na2HAs04 90 80 70 60 50 40 30 20 10 20 40 60 80 100 Temperature Figure 1: Solubility of different salts in water and different temperatures. Solubility (g per 100g water)
Step by Step Solution
3.45 Rating (148 Votes )
There are 3 Steps involved in it
Solution Curve in which solubility increbier or decreaser irregularly uth temperatu re is c... View full answer

Get step-by-step solutions from verified subject matter experts


