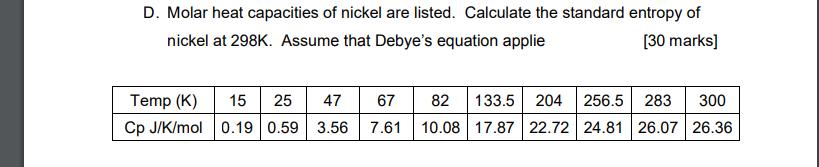
Question: D. Molar heat capacities of nickel are listed. Calculate the standard entropy of nickel at 298K. Assume that Debye's equation applie [30 marks] 15
D. Molar heat capacities of nickel are listed. Calculate the standard entropy of nickel at 298K. Assume that Debye's equation applie [30 marks] 15 25 47 67 Cp J/K/mol 0.19 0.59 3.56 204 256.5 283 10.08 17.87 22.72 24.81 26.07 26.36 Temp (K) 82 133.5 300 7.61
Step by Step Solution
There are 3 Steps involved in it
According to Debyes equation heat capacity of a sol... View full answer

Get step-by-step solutions from verified subject matter experts