Solve the inverse Laplace transform Questions: (stz)4} 35+2 2+25H0 Questions: (stz)4} 35+2 2+25H0
Fantastic news! We've Found the answer you've been seeking!
Question:
Solve the inverse Laplace transform
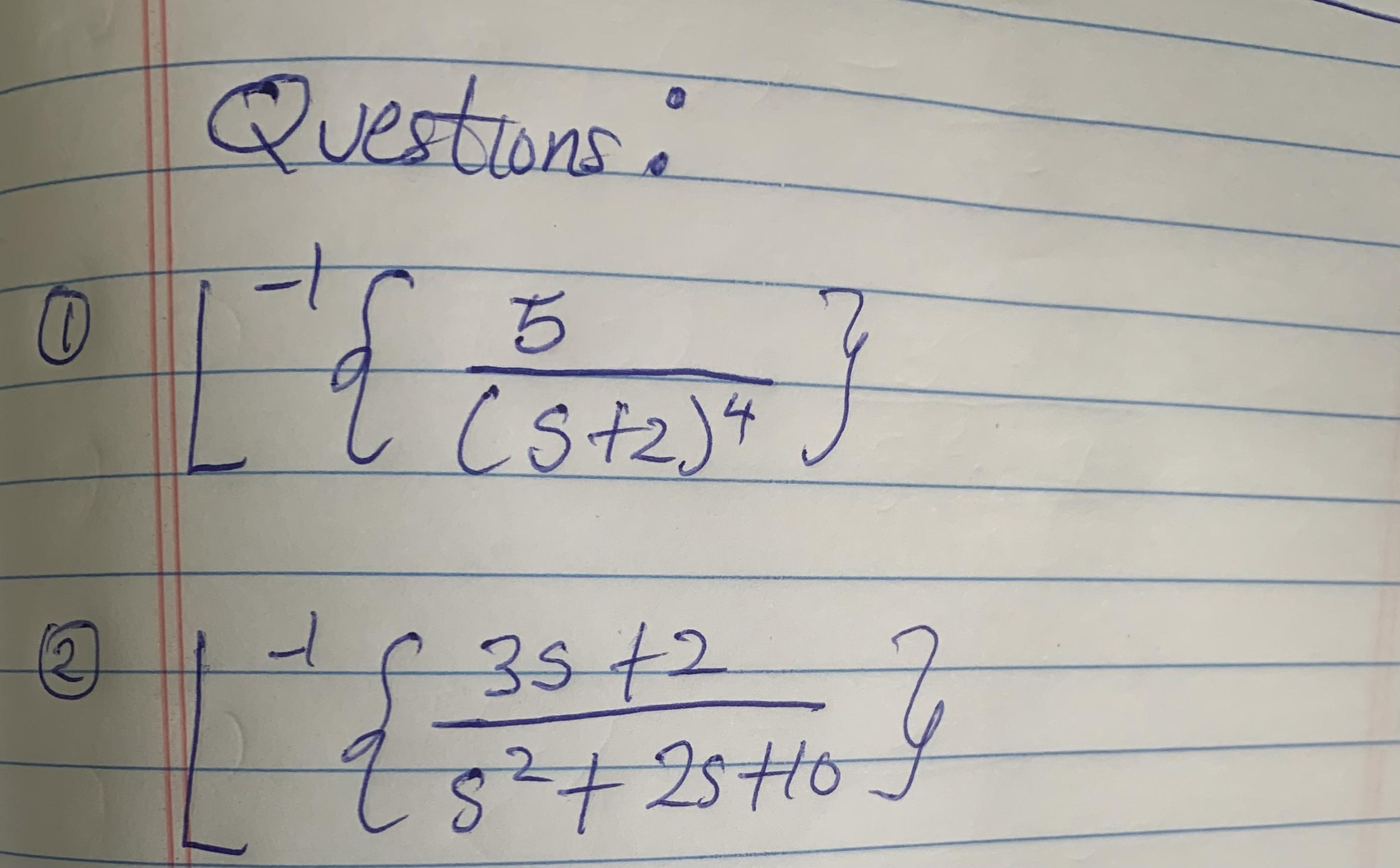
Transcribed Image Text:
Questions: (stz)4} 35+2 2+25H0 Questions: (stz)4} 35+2 2+25H0
Expert Answer:
Answer rating: 100% (QA)
S 24 We know if LE8 flt then LI Flsa e at flt ft eat L FIS1 ... View the full answer

Posted Date:
Students also viewed these mathematics questions
-
Solve the inverse Laplace transform 75-1 [ Cot 1) Cstz) Cst3, 25+10s Cs-23+5
-
Solve the following inverse Laplace transform and find k. Given 3 H(s) = s(s? + 25) Find the value of constant k if L-' {H(s)} = k(t u) sin (5u) du. Hence, find the inverse.
-
In the following problems we use the inverse Laplace transform and the relation between input and output of LTI systems. (a) The Laplace transform of the output of a system is find y 1 (t), assume it...
-
After reviewing the definition of Behavior Leadership Theory, explain what effect behavioral leadership theory has on traditional police in their role as a chief or any other leadership role in law...
-
This exercise requires the use of a computer package. The authors of the article Absolute Versus per Unit Body Length Speed of Prey as an Estimator of Vulnerability to Predation (Animal Behaviour...
-
On June 30, New Haven Companys work in Process inventory account showed a beginning balance of $29,400. The Materials Inventory account showed a beginning balance of $240,000. Production activity for...
-
You are holding a basketball while standing on a bathroom scale. You now throw the basketball directly upwards, and catch it on its way down. Describe how the reading on the scale changes from its...
-
Indicate the effects of the transactions listed in the following table on total current assets, current ratio, and net income. Use (+) to indicate an increase, (-) to indicate a decrease, and (0) to...
-
In the space provided, 150- to 350-word summary of your financial analysis from Part 2: Financial Ratio Calculations. Include the following in your summary: Make final recommendations as to whether...
-
Rainmaker Environmental Consultants is just finishing its second year of operations. The companys unadjusted trial balance at October 31, 2023, follows. Rainmaker prepares adjustments each October...
-
Visualize the least effective leader you know. Identify the traits motives, and personal characteristics in which that person might be deficient?
-
Explain the reasons for the choices you made. What compelled you to make those specific benefit choices?
-
How is a diversification strategy related to the sustainable growth and development of an organization?
-
If management treats employees well, pays them a fair wage, communicates with them, and ensures that they have a safe and healthy work environment, there is no need for a union. Do you agree or...
-
What changes do you think this might cause in the immediate task environment for a business operating within the financial service industry? Look at the Financial Times online for information.
-
What are the reasons for strategic planners to pay more attention to the importance of planning and implementing an effective pricing strategy in a competitive market?
-
Suppose the depositors do not believe that any bank is safe and hold their cash outside the banking system. Explain how the bank run would affect money supply. With an aid of the money demand and...
-
A line l passes through the points with coordinates (0, 5) and (6, 7). a. Find the gradient of the line. b. Find an equation of the line in the form ax + by + c = 0.
-
The latent heat of vaporization per unit mass of a pure substance at a given temperature, \(\lambda\), is defined as the difference in enthalpy between the saturated vapor and saturated liquid at the...
-
Calculate the absolute humidity, relative saturation, and dew-point temperature of a mixture of benzene-nitrogen at a pressure of \(1.5 \mathrm{bar}\) and temperature of \(320 \mathrm{~K}\) if the...
-
It is desired to dehumidify \(1.2 \mathrm{~m}^{3} / \mathrm{s}\) of air, available at \(311 \mathrm{~K}\) with a wet-bulb temperature of \(303 \mathrm{~K}\), to a wet-bulb temperature of \(288...

Study smarter with the SolutionInn App

