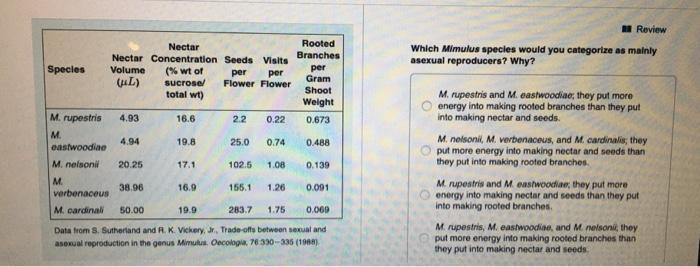
Species M. rupestris M. Nectar Nectar Concentration Seeds Visits eastwoodiae M. nelsoni M. Volume (L) 4.93...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Species M. rupestris M. Nectar Nectar Concentration Seeds Visits eastwoodiae M. nelsoni M. Volume (L) 4.93 4.94 20.25 38.96 (% wt of sucrose/ total wt) verbenaceus M. cardinali 50.00 16.6 19.8 25.0 0.74 102.5 1.08 0.139 155.1 1.26 0.091 283.7 1.75 0.069 Data from S. Sutherland and R. K. Vickery, Jr., Trade-offs between sexual and asexual reproduction in the genus Mimulus. Oecologia, 76330-335 (1988) 17.1 16.9 per per Flower Flower 19.9 2.2 0.22 Rooted Branches per Gram Shoot Weight 0.673 0.488 Review Which Mimulus species would you categorize as mainly asexual reproducers? Why? M. rupestris and M. eastwoodiae; they put more energy into making rooted branches than they put into making nectar and seeds. M. nelsonil, M. verbenaceus, and M. cardinalis, they put more energy into making nectar and seeds than they put into making rooted branches. M. rupestris and M. eastwoodiae; they put more energy into making nectar and seeds than they put into making rooted branches. M. rupestris, M. eastwoodiae, and M. nelsoni, they put more energy into making rooted branches than they put into making nectar and seeds. Species M. rupestris M. Nectar Nectar Concentration Seeds Visits eastwoodiae M. nelsoni M. Volume (L) 4.93 4.94 20.25 38.96 (% wt of sucrose/ total wt) verbenaceus M. cardinali 50.00 16.6 19.8 25.0 0.74 102.5 1.08 0.139 155.1 1.26 0.091 283.7 1.75 0.069 Data from S. Sutherland and R. K. Vickery, Jr., Trade-offs between sexual and asexual reproduction in the genus Mimulus. Oecologia, 76330-335 (1988) 17.1 16.9 per per Flower Flower 19.9 2.2 0.22 Rooted Branches per Gram Shoot Weight 0.673 0.488 Review Which Mimulus species would you categorize as mainly asexual reproducers? Why? M. rupestris and M. eastwoodiae; they put more energy into making rooted branches than they put into making nectar and seeds. M. nelsonil, M. verbenaceus, and M. cardinalis, they put more energy into making nectar and seeds than they put into making rooted branches. M. rupestris and M. eastwoodiae; they put more energy into making nectar and seeds than they put into making rooted branches. M. rupestris, M. eastwoodiae, and M. nelsoni, they put more energy into making rooted branches than they put into making nectar and seeds.
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below Answer First Optionp To answer this que... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these accounting questions
-
Describe the difference between sexual and asexual reproduction in zygomycetes. Why might asexual reproduction be more common than sexual reproduction?
-
Why do you think international trade volume has increased over time? In general, how are inefficient firms affected by the reduction in trade restrictions among countries and the continuous increase...
-
More than 75% of all species that have been described by biologists are insects. Of the approximately 2 million known species, only about 30,000 are aquatic in any life stage. The data in Table 1.4.1...
-
Axel Corporation acquires 100% of the stock of Wheal Company on December 31, Year 4. The following information pertains to Wheal Company on the date of acquisition: Book Value Fair Value Cash $...
-
Suppose you are an economic expert who works as an economic analyst in a small open economy (SOE) that presently has perfect financial capital mobility and no risk premium. The government of a large...
-
What is a framework? What are the internal control frameworks recognized globally by management, independent outside accountants/auditors, and internal audit professionals?
-
Describe the nature and purpose of an ethical wall.
-
Over the past year, M. D. Ryngaert & Co. has realized an increase in its current ratio and a drop in its total assets turnover ratio. However, the companys sales, quick ratio, and fixed assets...
-
Exercise 13-3 (Algo) Sourcing Decisions [LO13-3] Troy Engines, Limited, manufactures a variety of engines for use in heavy equipment. The company has always produced all of the parts for its engines,...
-
Julio and Milania are owners of Falcons Corporation, an S corporation. They each own 50 percent of Falcons Corporation. In year 1, Julio and Milani received distributions of $20,000 and $10,000,...
-
You have purchased a put. This one is currently strongly out of the money. What about the price of the underlying and the strike? The underlying asset suddenly becomes much more volatile and its...
-
Given the information in Problems 41-44, find the equation for the least squares line as well as the correlation coefficient. \(n=10, \Sigma x=57, \Sigma y=1, \Sigma x y=-13, \Sigma x^{2}=353\),...
-
JOURNAL PROBLEM The graph shown in Figure 14.42 is from the February 1991 issue of Scientific American. If the curve in the middle is a standard normal curve, describe the variance of the upper curve...
-
Explain the principle of tandeed in Islam.
-
In Problems 7-18, a sample of paired data gives a linear correlation coefficient \(r\). In each case, use Table 14.10 to determine whether there is a significant linear correlation. Table 14. 10...
-
The national debt is of growing concern in the United States. It reached \$1 billion in 1916 during World War I and climbed to \(\$ 278\) billion by the end of World War II. It reached its first...
-
A racecar is racing along a circular track. The car starts at the 3-o'clock position and travels CCW along the track. The car is constantly 9 feet from the center of the race track and travels at a...
-
Is that Yelp review real or fake? The article A Framework for Fake Review Detection in Online Consumer Electronics Retailers (Information Processing and Management 2019: 12341244) tested five...
-
The concentration of gold in seawater has been reported to be between 5 ppt (parts per trillion) and 50 ppt. Assuming that seawater contains 13 ppt of gold, calculate the number of grams of gold...
-
The contour representation of one of the orbital's for the n = 3 shell of a hydrogen atom is shown below. (a) What is the quantum number l for this orbital? (b) How do we label this orbital? (c) How...
-
Lauryl alcohol is obtained from coconut oil and is used to make detergents. A solution of 5.00 g of lauryl alcohol in 0.100 kg of benzene freezes at 4.1 oC. What is the approximate molar mass of...
-
Compare the air-standard cycle thermal efficiencies of an Otto cycle obtained by calculating the state points around the cycle with the value obtained from Eqn (3.16) for an 'engine' operating with a...
-
Compare the air-standard cycle thermal efficiencies of a diesel cycle obtained by calculating the state points around the cycle with the value obtained from Eqn (3.20) for an 'engine' operating with...
-
Recalculate the Otto cycle in P16.5 using EQUIL2 to evaluate the conditions around the cycle. Do these calculations both with and without considering dissociation. How has the use of more accurate...

Study smarter with the SolutionInn App


