Question: TABLE 6.8 Molecule Total # Valence Electrons Lewis Structure Number of Bonding Electrons clouds on one carbon atom Number of Nonbonding Electron Pairs on
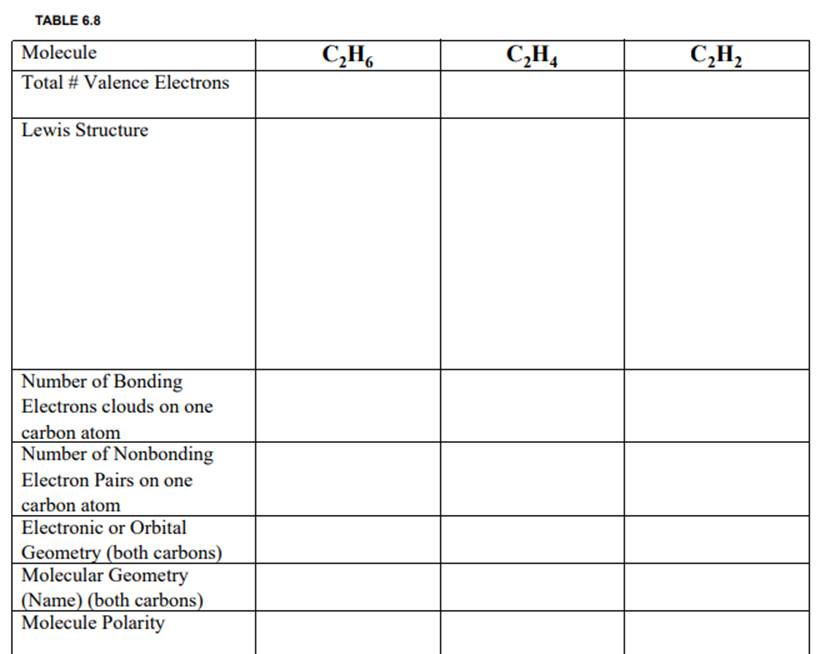
TABLE 6.8 Molecule Total # Valence Electrons Lewis Structure Number of Bonding Electrons clouds on one carbon atom Number of Nonbonding Electron Pairs on one carbon atom Electronic or Orbital Geometry (both carbons) Molecular Geometry (Name) (both carbons) Molecule Polarity CH6 CH4 CH
Step by Step Solution
3.48 Rating (148 Votes )
There are 3 Steps involved in it
TABLE 68 Molecule CH CH CH Total Valence Electrons ... View full answer

Get step-by-step solutions from verified subject matter experts


