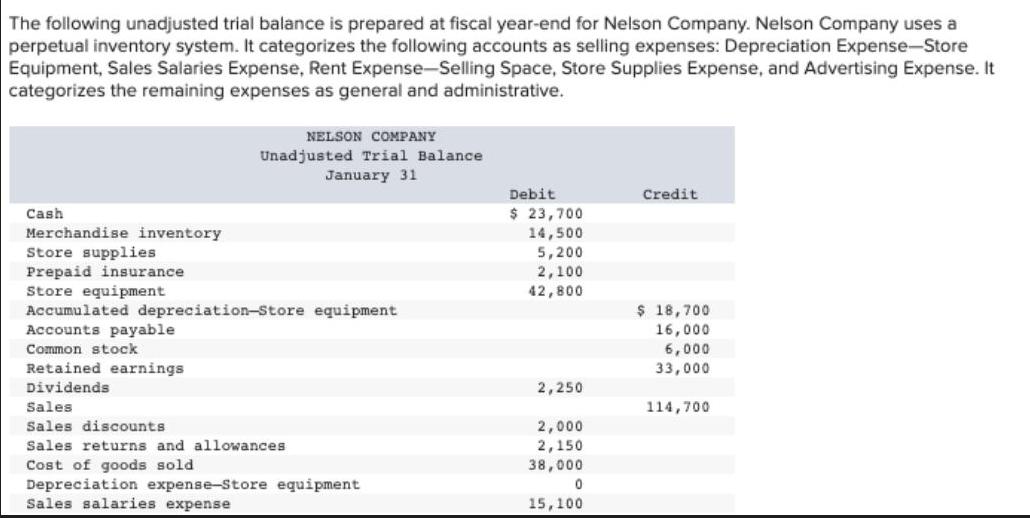
The following unadjusted trial balance is prepared at fiscal year-end for Nelson Company. Nelson Company uses...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
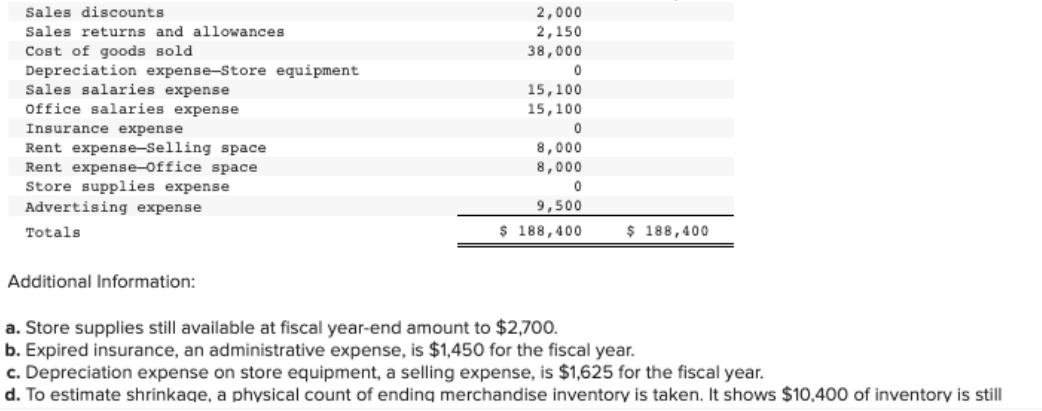
The following unadjusted trial balance is prepared at fiscal year-end for Nelson Company. Nelson Company uses a perpetual inventory system. It categorizes the following accounts as selling expenses: Depreciation Expense-Store Equipment, Sales Salaries Expense, Rent Expense-Selling Space, Store Supplies Expense, and Advertising Expense. It categorizes the remaining expenses as general and administrative. Cash Merchandise inventory Store supplies Prepaid insurance Store equipment NELSON COMPANY Unadjusted Trial Balance January 31 Accumulated depreciation-Store equipment Accounts payable Common stock Retained earnings Dividends Sales Sales discounts Sales returns and allowances Cost of goods sold Depreciation expense-Store equipment Sales salaries expense Debit $ 23,700 14,500 5,200 2,100 42,800 2,250 2,000 2,150 38,000 0 15,100 Credit $ 18,700 16,000 6,000 33,000 114,700 Sales discounts Sales returns and allowances. Cost of goods sold Depreciation expense-Store equipment Sales salaries expense Office salaries expense Insurance expense Rent expense-Selling space Rent expense-Office space Store supplies expense Advertising expense Totals Additional Information: 2,000 2,150 38,000 0 15,100 15,100 0 8,000 8,000 0 9,500 $ 188,400 $ 188,400 a. Store supplies still available at fiscal year-end amount to $2,700. b. Expired insurance, an administrative expense, is $1,450 for the fiscal year. c. Depreciation expense on store equipment, a selling expense, is $1,625 for the fiscal year. d. To estimate shrinkage, a physical count of ending merchandise inventory is taken. It shows $10,400 of inventory is still The following unadjusted trial balance is prepared at fiscal year-end for Nelson Company. Nelson Company uses a perpetual inventory system. It categorizes the following accounts as selling expenses: Depreciation Expense-Store Equipment, Sales Salaries Expense, Rent Expense-Selling Space, Store Supplies Expense, and Advertising Expense. It categorizes the remaining expenses as general and administrative. Cash Merchandise inventory Store supplies Prepaid insurance Store equipment NELSON COMPANY Unadjusted Trial Balance January 31 Accumulated depreciation-Store equipment Accounts payable Common stock Retained earnings Dividends Sales Sales discounts Sales returns and allowances Cost of goods sold Depreciation expense-Store equipment Sales salaries expense Debit $ 23,700 14,500 5,200 2,100 42,800 2,250 2,000 2,150 38,000 0 15,100 Credit $ 18,700 16,000 6,000 33,000 114,700 Sales discounts Sales returns and allowances. Cost of goods sold Depreciation expense-Store equipment Sales salaries expense Office salaries expense Insurance expense Rent expense-Selling space Rent expense-Office space Store supplies expense Advertising expense Totals Additional Information: 2,000 2,150 38,000 0 15,100 15,100 0 8,000 8,000 0 9,500 $ 188,400 $ 188,400 a. Store supplies still available at fiscal year-end amount to $2,700. b. Expired insurance, an administrative expense, is $1,450 for the fiscal year. c. Depreciation expense on store equipment, a selling expense, is $1,625 for the fiscal year. d. To estimate shrinkage, a physical count of ending merchandise inventory is taken. It shows $10,400 of inventory is still
Expert Answer:
Answer rating: 100% (QA)
1 Working Notes are prepared for Adjusting Entries a Supplies Expenses Supplies Available Supplies Ending 5200 2700 2500 b Cost of Goods Sold Merchandise Inventory Available Merchandise Inventory Endi... View the full answer

Related Book For 

Fundamental Accounting Principles
ISBN: 978-1259536359
23rd edition
Authors: John Wild, Ken Shaw, Barbara Chiappett
Posted Date:
Students also viewed these accounting questions
-
The following unadjusted trial balance is prepared at fiscal year-end for Nelson Company. Rent expense and salaries expense are equally divided between selling activities and general and...
-
The following unadjusted trial balance is prepared at fiscal year-end for Foster Products Company. Rent expense and salaries expense are equally divided between selling activities and general and...
-
How the Gram-Schmidt process is related to the Gaussian elimination method?
-
On January 1, 20X7, CP Co. (a Canadian company) purchased 80% of SF Co. (a U.S. company) at a cost of US$50,000. The book values of SF's net assets were equal to their fair market values on this date...
-
Len Thomas, the accountant for Lancaster Consulting, prepared the worksheet shown on the next page on a computer spreadsheet but has lost much of the data. The only particular item Thomas can recall...
-
Varnay Ltd is a manufacturing company which prepares annual financial statements to 31 December. In November 2017, the company announced a plan to close down one of its manufacturing operations and...
-
The force transmissibility of a damped single-degree-of-freedom system with base motion is given by Eq. (9.106): \[T_{f}=\frac{F_{t}}{k Y}=r^{2}\left\{\frac{1+(2 \zeta r)^{2}}{\left(1-r^{2}...
-
The following labor hours data have been collected for a nanotechnology project for periods 1 through 6. Compute the SV, CV, SPI, and CPI for each period. Plot the EV and the AC on the summary graph...
-
Kwan acquired a warehouse for business purposes on August 30, 2002. The building cost $420,000. Kwan took $227,600 of depreciation on the building, and then sold it for $500,000 on July 1, 2021....
-
The following information is available for the preparation of the governmentwide financial statements for the city of Southern Springs as of April 30, 2020: From the preceding information, prepare,...
-
3.1 Determine the 75%, 90%, and 95% response time for each of the systems given (assume zero initial conditions): 2P + 8P + 8P = 2U(t) a. b. 5y +5y = U(t)
-
The Tasha Corporation needs a new pipe-boring machine that can be purchased for $10,000.The machine will be depreciated using MACRS with a 5-year class.Tasha will use the machine for 5 years and then...
-
Assumptions Yearly Inflation Rate: 3% Funds and 10-year returns shown below in table and graph format (the 10th year being last year's returns): 1 2 3 4 5 6 7 8 9 10 Crimson 200 Mercury 500 Bonds...
-
The table below provides the grid coordinates and the estimated demand for a set of cities that your company wishes to connect through a centrally located distribution center. Where would you locate...
-
Classify the singularities of this algebraic plane curve. y = x (x 1)(x 2) - Classify the singularities of this algebraic plane curve. - y = (x 2)(x 4)(x 6)
-
Find an algebraic expression equal to sec (sin (You may assume that < sin (5x 4) < ) Provide your answer below: sec(sin (5x = 4)) = (5x-4)).
-
Working Conditions Ratings Dallas Plant San Antonio 3 4 6 4 3 4 5 3 4 5 3 5 4 3 4 4 4 4 3 4 5 5 4 5 3 4 6 4 2 3 1 3 4 4 5 4 3 4 6 4 4 5 4 5 3 3 4 4 4 6 4 4 3 6 3 4 4 5 4 4 3 5 1 4 2 4 3 2 3 1 4 3 2 4...
-
Suppose the S&P 500 futures price is 1000, = 30%, r = 5%, = 5%, T = 1, and n = 3. a. What are the prices of European calls and puts for K = $1000? Why do you find the prices to be equal? b. What...
-
Locate "Note 16," which reports Google's geographical segments from its 2015 annual report (sec.gov/Archives/edgar/data/1288776/000165204416000012/ goog10-k2015.htm). Identify its geographical...
-
Forten Company, a merchandiser, recently completed its calendar-year 2017 operations. For the year, (1) all sales are credit sales, (2) all credits to Accounts Receivable reflect cash receipts from...
-
Prepare any necessary adjusting entries at December 31, 2017, for Piper Company's year-end financial statements for each of the following separate transactions and events. 1. Piper Company records a...
-
A system, consisting of \(2 \mathrm{~mol}\) of \(\mathrm{N}_{2}, 5 \mathrm{~mol}\) of \(\mathrm{H}_{2}\) and \(2 \mathrm{~mol}\) of \(\mathrm{NH}_{3}\) initially, is undergoing the following...
-
The following reaction takes place in a system consisting of \(3 \mathrm{~mol} \mathrm{CH}_{4}, 5 \mathrm{~mol} \mathrm{H}_{2} \mathrm{O}\), \(1 \mathrm{~mol} \mathrm{CO}\) and \(4 \mathrm{~mol}...
-
The study of thermodynamics enables us to understand (a) Whether the transformation of energy is feasible or not (b) To what extent the transformation will take place (c) In which direction the...

Study smarter with the SolutionInn App


