Question: The heat pump cycle shown in Fig. P6.53 operates at steady state and provides energy by heat transfer at a rate of 15 kW
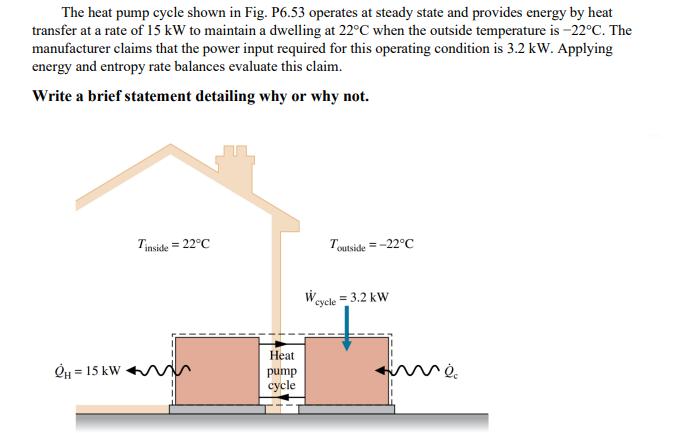
The heat pump cycle shown in Fig. P6.53 operates at steady state and provides energy by heat transfer at a rate of 15 kW to maintain a dwelling at 22C when the outside temperature is -22C. The manufacturer claims that the power input required for this operating condition is 3.2 kW. Applying energy and entropy rate balances evaluate this claim. Write a brief statement detailing why or why not. OH = 15 kW Tinside = 22C Heat pump cycle W Toutside=-22C cycle = 3.2 kW me
Step by Step Solution
3.42 Rating (155 Votes )
There are 3 Steps involved in it
Apply energy Qc wc OH 0 Qc 3215 0 Qc 118 kW By entrop... View full answer

Get step-by-step solutions from verified subject matter experts


