At midnight, a hospital emergency room (E.R.) has 3 patients. The rate that new patients are...
Fantastic news! We've Found the answer you've been seeking!
Question:

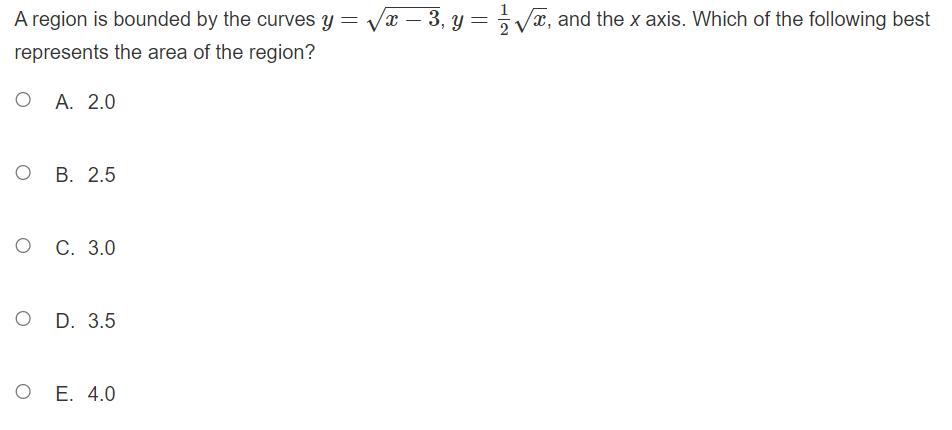
Transcribed Image Text:
At midnight, a hospital emergency room (E.R.) has 3 patients. The rate that new patients are admitted to the E.R. over the next 24 hours is modeled by the function A (t) =(768 +23t-t²). The rate that E.R. patients are released over the same time period is modeled by the function 65 R (t) = (390+41t – t²). The units for both A (t) and R (t) are in people per hour. Approximately how many patients are in the E.R. at the end of the 24-hour time period? OA. 3 O B. 5 O C. 7 O D. 9 OE. 11 A region is bounded by the curves y = √√x - 3, y = √, and the x axis. Which of the following best represents the area of the region? OA. 2.0 O B. 2.5 O C. 3.0 D. 3.5 OE. 4.0 At midnight, a hospital emergency room (E.R.) has 3 patients. The rate that new patients are admitted to the E.R. over the next 24 hours is modeled by the function A (t) =(768 +23t-t²). The rate that E.R. patients are released over the same time period is modeled by the function 65 R (t) = (390+41t – t²). The units for both A (t) and R (t) are in people per hour. Approximately how many patients are in the E.R. at the end of the 24-hour time period? OA. 3 O B. 5 O C. 7 O D. 9 OE. 11 A region is bounded by the curves y = √√x - 3, y = √, and the x axis. Which of the following best represents the area of the region? OA. 2.0 O B. 2.5 O C. 3.0 D. 3.5 OE. 4.0
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these mathematics questions
-
There is only one value of the equilibrium constant for a particular system at a particular temperature, but there are an infinite number of equilibrium positions. Explain.
-
Calculate the KP for this reaction at 298 K if the Keq = 1.76 103. 3O2 (g) + 2O3(g)
-
Calculate the KP for this reaction at 310 K if the Keq = 6.22 103. 4NO2 (g) + O2 (g) + 2N2O5 (g)
-
Foreman Publishing Companys income for the most recent quarter was $500,000, and the average net book value of assets during the quarter was $1.5 million. If the company has a required rate of return...
-
How would promotional mix decisions change for a small business that is expanding into a foreign market?
-
1. A company has three bonds they've issuedone's with coupon rates of 4.5%, 4.75%, and 5.2%. Together, the bonds are listed on their SFP at $300 million. The three have been priced in the market to...
-
How might an organization estimate the direct and indirect benefits of a new HRIS that decreases the time required by employees to complete transactions of the HR department through the...
-
In college basketball, a turnover is defined as losing possession of the basketball to the opposing team. During a recent basketball season, the Villanova Wildcats averaged 13 turnovers per game....
-
The Clarkson Coffee Company produces two grades of coffee, the Superb blend and the Run-of-the-Mill blend. Three types of coffee beans are combined to produce these blends. The availability and cost...
-
The following financial statements and additional information are reported. IKIBAN INC. Comparative Balance Sheets June 30, 2017 and 2016 2017 2016 Assets Cash $ 101,500 $ 54,000 Accounts receivable,...
-
Lopez Sales Company had the following balances in its accounts on January 1, Year 2 Cash Merchandise Inventory Land Common Stock Retained Earnings $ 53,000 33,000 93,000 73,000 106,000 Lopez...
-
5- (25pts)- Consider the signal m(t) = cos(27ft) + 2 cos(27ft) fi=3kHz, f=5kHz. m(t) VSB modulates a carrier with frequency f=50kHz. The lowpass filter Ho(f) is given as |H/j -6 0.5 10.5 11 2 4 6...
-
RSE, Inc. is a US producer of recreational sports equipment. Suppose that you, as the CFO of RSE, Inc, Inc., believe the growth potential for some of sports equipment, particularly tennis and squash...
-
= Three parallel plates having the same surface area A, are positioned as shown in the figure. The upper two plates are moving at V and V velocities while the bottom plate is fixed. A fluid having...
-
On January 1, 1980, your favorite uncle John turned 43 and started saving for his retirement. He invested $18,894.75 each year on January 1 until he turned 62 on 1/1/1999 (20 total deposits). Over...
-
Suppose that Amy has the following value function: v(w) = w.3/4 when w > 0 = -2(-w) 3/4 when w < 0 where x is the realized outcome relative to the reference point. Assume that the my's reference...
-
Find the inverse for each function. Then graph the function and its inverse. If necessary, restrict the domain of f(x) so that the inverse is a function. 24-29. See Ch. 5 Answer Appendix. 24. f(x) =...
-
Suppose a population of bacteria doubles every hour, but that 1.0 x 106 individuals are removed before reproduction to be converted into valuable biological by-products. Suppose the population begins...
-
Write the equilibrium-constant expression for the equilibrium The table that follows shows the relative mole percentages of CO2(g) and CO(g) at a total pressure of 1 atm for several temperatures....
-
Calculate the percent ionization of propionic acid (C2H5COOH) in solutions of each of the following concentrations (Ka is given in Appendix D): (a) 0.250 M, (b) 0.0800 M, (c) 0.0200 M.
-
Calculate the pH at the equivalence point in titrating 0.100 M solutions of each of the following with 0.080 MNaOH: (a) hydrobromic acid (HBr), (b) chlorous acid (HClO2), (c) benzoic acid (C6H5COOH).
-
Diehl Cleaners has the following statement of financial position items. Instructions Classify each item as an asset, liability, or equity. Analyze the effect of transactions. Accounts payable Cash...
-
Erin Danielle, the bookkeeper for Liverpool Ltd., has been trying to determine the correct statement of financial position for the company. The companys statement of financial position is shown...
-
Which of the following is true? a. Financial frauds have not occurred in U.S. companies because GAAP has detailed accounting and disclosure requirements. b. Transaction analysis is basically the same...

Study smarter with the SolutionInn App


