The mass fraction of O2, N2, and CO2 which are mixed inside a rigid tank at...
Fantastic news! We've Found the answer you've been seeking!
Question:
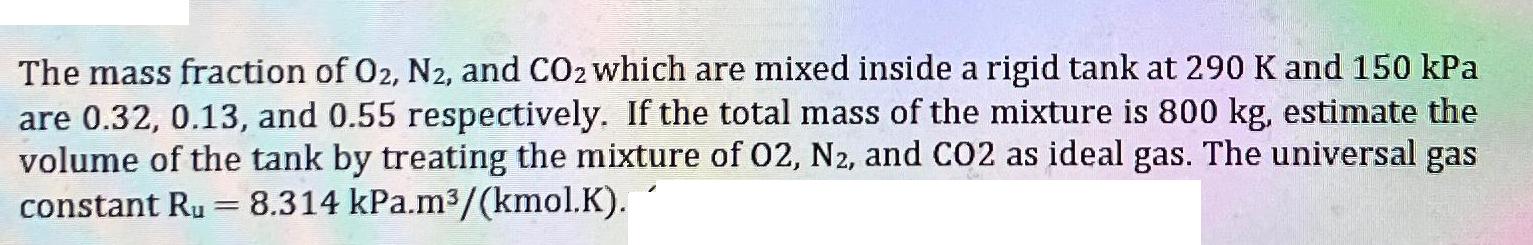
Transcribed Image Text:
The mass fraction of O2, N2, and CO2 which are mixed inside a rigid tank at 290 K and 150 kPa are 0.32, 0.13, and 0.55 respectively. If the total mass of the mixture is 800 kg, estimate the volume of the tank by treating the mixture of 02, N2, and CO2 as ideal gas. The universal gas constant Ru 8.314 kPa.m³/(kmol.K). = The mass fraction of O2, N2, and CO2 which are mixed inside a rigid tank at 290 K and 150 kPa are 0.32, 0.13, and 0.55 respectively. If the total mass of the mixture is 800 kg, estimate the volume of the tank by treating the mixture of 02, N2, and CO2 as ideal gas. The universal gas constant Ru 8.314 kPa.m³/(kmol.K). =
Expert Answer:
Answer rating: 100% (QA)
To estimate the volume of the tank with a mixture of O2 N2 and CO2 gases we can use the ideal gas la... View the full answer

Related Book For 

Thermodynamics for Engineers
ISBN: ?978-1133112860
1st edition
Authors: Kenneth A. Kroos, Merle C. Potter
Posted Date:
Students also viewed these accounting questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
A gas mixture of 4 kg of O2, 6 kg of N2, and 8 kg of CO2 is contained at a pressure of 120 kPa and 20 8 C in a rigid container. Calculate: i) The mass fraction of each component ii) The mole fraction...
-
The following data represent the muzzle velocity (in feet per second) of rounds red from a 155-mm gun. For each round, two measurements of the velocity were recorded using two different measuring...
-
A baseball "diamond" (FIGURE 3-38) is a square with sides 90 ft in length. If the positive x axis points from home plate to first base, and the positive y axis points from home plate to third base,...
-
Rose Company has a relevant range of production between 10,000 and 25,000 units. The following cost data represents average cost per unit for 15,000 units of production. Using the cost data from Rose...
-
An ergodic real-valued random process \(U(t)\) with autocorrelation function \(\Gamma_{U}(\tau)=\left(N_{0} / 2 ight) \delta(\tau)\) is applied to the input of a linear, time-invariant filter with...
-
On January 1, 2016, Ballieu Company leases specialty equipment with an economic life of 8 years to Anderson Company. The lease contains the following terms and provisions: The lease is noncancelable...
-
Suppose that there is a bond with 12 years left to maturity. The face value of the bond is $1,000. Coupon rate is 15%, and YTM is 12%. Coupons are paid semiannually. What is the price that you are...
-
Capstone Case: Sunrise Bakery Expansion The Sunrise Bakery Corporation was originally founded in Houston, TX in 1991 by Griffin Harris, who currently serves as the company's Chief Executive Officer....
-
A. Shelley Construction began operations in 2020 and appropriately used the completed-contract method in accounting for its long-term construction contracts. They prepared the following information:...
-
A sex offender is convicted and sentenced to a long jail term. Say 10 years. After serving their sentence rather than being released they are civilly committed and remain incarcerated at a mental...
-
Antonio believes he has been misled by his employer, MiraCosta College. He decides to sue. He knows you have recently taken BUS140 and asks what you think about going to court. Would you advise him...
-
Jamie Jones is a resident at Miller's Retirement Center. She has dementia. Some days she seems without ailment. Dr. Smith needs her permission for a minor surgery. Is there any way to get Ms. Jones'...
-
Harry lends his car to his friend Alice for the month of January while she is away on holiday overseas. a) Explain who the owner of the car is during this time. b) If Alice allows her younger sister...
-
Jeanne Dunne, a bank teller, provided a description of a bank robber "moments" following the robbery. The robber was identified as an African American with dark skin, a wide nose, and medium build....
-
Pina Construction Ltd. reported the following information related to its shareholders' equity on January 1, 2021, the first day of its fiscal year. $0.50-noncumulative preferred shares, unlimited...
-
1. True or False. Pitfalls to consider in a statistical test include nonrandom samples, small sample size, and lack of causal links. 2. Because 25 percent of the students in my morning statistics...
-
An automobile has a gas mileage of 30 miles/gal when traveling at 60 mph on a flat road with no head wind. Gas contains about 21,000 Btu/lbm with a density of 45 lbm/ft3. Estimate the efficiency of...
-
The COP of the gas air-conditioning cycle of Problem 10.7 is nearest: (a) 0.29 (b) 1.05 (c) 1.34 (d) 1.98 Heat exchange| | 100 C Gas net comD Turbine o C 80 kPa Heat exchanger
-
Apply Eq. 7.7 directly to Eqs. 7.1 and 7.2 and derive the relations of Eqs. 7.15 and 7.16. Eq. 7.1 du = Tds - Pdv Eq. 7.2 dh = Tds + vdP Eq. 7.7 dz = (z/x)y dx + (z/y)x dy Eq. 7.15 (u/s)v = T and...
-
Briefly explain the difference between accounting, finance, and engineering economics. Try to put the concepts in your own (or your team's) words and compare the concepts where appropriate.
-
What are the two key financial objectives in the management of a company? How can a focus on these objectives create ethical dilemmas?
-
Among your colleagues in class, identify a term or phrase italicized in this chapter that you think is the most significant from your reading. Absent team consensus, then just provide your...

Study smarter with the SolutionInn App


