The rate of hydrolysis, K, of methyl acetate is 4 times faster at 25 C than...
Fantastic news! We've Found the answer you've been seeking!
Question:
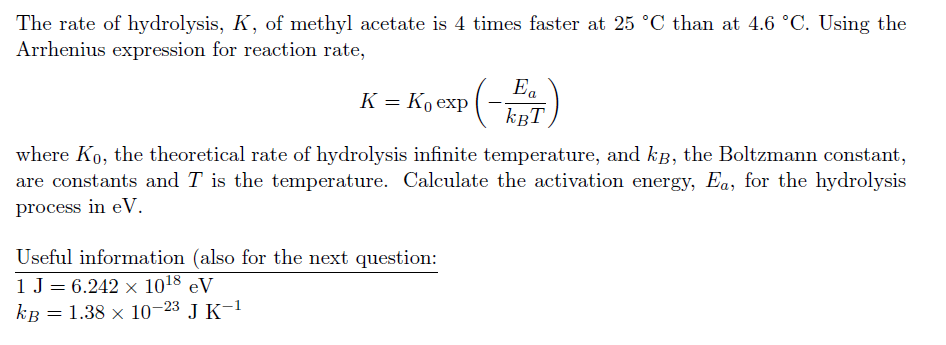
Transcribed Image Text:
The rate of hydrolysis, K, of methyl acetate is 4 times faster at 25 C than at 4.6 C. Using the Arrhenius expression for reaction rate, Ea K = K exp(-4) KBT where Ko, the theoretical rate of hydrolysis infinite temperature, and k, the Boltzmann constant, are constants and T is the temperature. Calculate the activation energy, Ea, for the hydrolysis process in eV. Useful information (also for the next question: 1 J = 6.242 108 eV kB = 1.38 x 10-23 J K- The rate of hydrolysis, K, of methyl acetate is 4 times faster at 25 C than at 4.6 C. Using the Arrhenius expression for reaction rate, Ea K = K exp(-4) KBT where Ko, the theoretical rate of hydrolysis infinite temperature, and k, the Boltzmann constant, are constants and T is the temperature. Calculate the activation energy, Ea, for the hydrolysis process in eV. Useful information (also for the next question: 1 J = 6.242 108 eV kB = 1.38 x 10-23 J K-
Expert Answer:
Answer rating: 100% (QA)
The given question is a typical application of the Arrhenius equation which describes the temperatur... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Consider a 2-m-long wire of thermal diffusivity a=1 subjected to initial temperature 3x(2-x) and boundary temperature 1 and t. The governing equation is: = a at subject to T(0,t) = 1,T(2,t) =t and...
-
After watching the presentation titled The Sacred/Secular Divide, interact with your classmates by discussing the following questions: How does the tendency to push religion away from the public...
-
Suppose that in any given year the number of cases can be reduced by 25% instead of 20%. a. How long will it take to reduce the number of cases to 1000? b. How long will it take to eradicate the...
-
Mrs. Clarks Foods was an Iowa company engaged in the business of distributing juice beverages. International Suntrade and Miller & Smith Foods were Canadian companies that acted as brokers...
-
CV exercises. The Doral Company manufactures and sells pens. Currently, 5,000,000 units are sold per year at$0.50 per unit. Fixed costs are $900,000 per year. Variable costs are $0.30 per unit....
-
Brand X copier advertises that its copiers run 23% longer between service calls than its competitor. If Brand X copiers run 65,000 copies between services, how many copies would the competitor run?...
-
Use Solver to create Answer and Sensitivity Reports for question 15 at the end of Chapter 2 and answer the following questions: a. How much excess wiring and testing capacity exists in the optimal...
-
You wish to design an X-band imaging microwave radiometer for a speed craft at 385 km(ground speed is 7.24 km/sec? a) Determine the frequency. What is a wavelength in mm? b) What is antenna diameter...
-
The role and importance of quality service, hospitality, and guest services within the hotel Industry Hilton Worldwide, Briefly introduce your chosen hotel location Identify the hotel's main...
-
About consumption. to. Describes what the permanent income hypotheses and the Life Cycle hypothesis consist of. b. If so, what are the similarities and differences between the life cycle and...
-
Explain the system approach associated with the Covid 19 case. How does an open system organization receive information from the Covid 19 case, starting with the input, throughput and output...
-
Define brand association and explain its importance to marketing
-
How does the criminal justice organization/agency support the general goals of criminal justice?
-
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for AgCl(s) is -916 kJ/mol and the hydration enthalpy (dissolution of gaseous ions into water) is 850 kJ/mol. How much heat...
-
X-1 Find the domain of the function f(x) : x 1 2 - O (-00, -1) U (-1, ) O (-00, 1) U (1, ) O -00, -1) U (-1, 1) U (1, 0) O (- 1, 1)
-
A reversible liquid-phase isomerization A B is carried out isothermally in a 1000-gal CSTR. The reaction is second order in both the forward and reverse directions. The liquid enters at the top of...
-
The irreversible gas-phase dimerization 2A A2 is carried out at 8.2 atm in a stirred contained-solids reactor to which only pure A is fed. There are 40 g of catalyst in each of the four spinning...
-
Using a negative step tracer input, Cholette and Cloutier (Can. J. Chem. Eng., 37, 107 (1959)) studied the RTD in a tank for different stirring speeds. Their tank had a 30-in. diameter and a fluid...
-
With regard to genotypes, what is a true-breeding organism?
-
With regard to biological evolution, which of the following statements is incorrect? Explain why. A. During its lifetime, an animal evolves to become better adapted to its environment. B. The process...
-
Marfan syndrome is a rare inherited human disorder characterized by unusually long limbs and digits plus defects in the heart (especially the aorta) and the eyes, among other symptoms. Following is a...

Study smarter with the SolutionInn App


