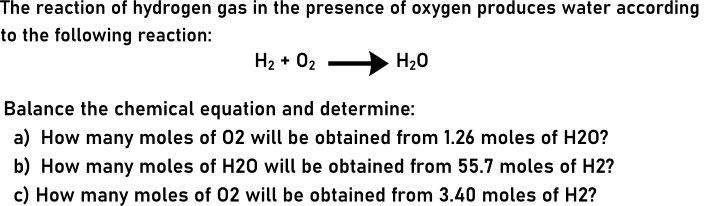
The reaction of hydrogen gas in the presence of oxygen produces water according to the following...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The reaction of hydrogen gas in the presence of oxygen produces water according to the following reaction: H2 + 02 H20 Balance the chemical equation and determine: a) How many moles of 02 will be obtained from 1.26 moles of H20? b) How many moles of H20 will be obtained from 55.7 moles of H2? c) How many moles of 02 will be obtained from 3.40 moles of H2? The reaction of hydrogen gas in the presence of oxygen produces water according to the following reaction: H2 + 02 H20 Balance the chemical equation and determine: a) How many moles of 02 will be obtained from 1.26 moles of H20? b) How many moles of H20 will be obtained from 55.7 moles of H2? c) How many moles of 02 will be obtained from 3.40 moles of H2?
Expert Answer:
Answer rating: 100% (QA)
solution The Reachion of Hydrogm gar in the presence of oxygen Pro... View the full answer

Posted Date:
Students also viewed these chemistry questions
-
How many moles of Al2O3 will be produced when 23.9 g of H2O are reacted according to this chemical equation? 2AlCl3 + 3H2O() ( Al2O3 + 6HCl(g)
-
The rate constant for the reaction of hydrogen with iodine is 2.45 10 4 M 1 s 1 at 302 C and 0.950 M 1 s 1 at 508 C. a. Calculate the activation energy and Arrhenius preexponential factor for this...
-
The information below gives the data for the reaction of hydrogen with iodine at 500C. H 2 (g) + I 2 (g) 2HI(g) The table shows the initial partial pressures and the partial pressures at equilibrium...
-
Consider a country that imports steel and exports automobiles. The value per unit of cars exported is measured in units of thousands of dollars per car by the random variable X. The value per unit of...
-
The National Oceanographic Data Center provides useful data for vacation planning. Below are listed beach temperatures in the month of July for various U.S. coastal areas. Using the 0.05 level of...
-
Quality Cost Classification Required Classify each of the following costs into one of the four quality cost (i.e., COQ) categories: 1. Materials, labor, and overhead costs of scrapped units. 2....
-
Describe the differences between civil law and criminal law. What is the purpose of criminal law? What is the purpose of civil law?
-
Sunset Construction, Inc. is a home builder in Arizona. Sunset uses a job order costing system in which each house is a job. Because it constructs houses, the company uses an account titled...
-
QUESTION FOUR The increasing demand in risk management on the part of organizations has been identified as a general societal trend emphasizing public accountability and responsibility. State and...
-
T. J. Patrick is a young, successful industrial designer in Portland, Oregon, who enjoys the excitement of commodities speculation. T. J. has been dabbling in commodities since he was a teen-ager-he...
-
What do you know about Stephen King? If you don't know anything about him, take a minute to look him up so that you have some context for his essay. In his opening, Stephen King states that the...
-
The world comprises two countries, A and B. There is only one good, whose price is normalized to 1; hence nominal and real measures coincide. These two countries differ with respect to their...
-
Texxon Corporation issued $500,000 of 8%, 10-year bonds. Interest is paid semiannually. Assume that the market rate for similar investments is 6 percent. What is the semi-annual cash payment amount...
-
The FIFA World Cup Qatar 2022 will be played from 20 November to 18 December in Qatar. The next 2026 tournament will be held in the United States, Mexico, and Canada. This is the biggest sports event...
-
Comparison rates in credit advertisements for fixed term credit for personal domestic household purposes must be made available. Select one: True False
-
The fact that the value of a good is subjective means that O the value of a good is equal to the labor used to produce it. O voluntary trade makes both parties better off. people do not look at...
-
Explain what is meant by vicarious liability and when it is available?
-
From output of your computer software, locate (a) the sum of squared errors, (b) the \(F\)-value for the overall significance of a regression model, (c) the estimated covariance matrix for the least...
-
Obtain restricted least squares estimates that include nonsample information in the estimation procedure.
-
Use the \(F\)-test to test single null hypotheses or joint null hypotheses.

Study smarter with the SolutionInn App

