The reaction of nitrogen gas in the presence of hydrogen produces ammonia according to the following...
Fantastic news! We've Found the answer you've been seeking!
Question:
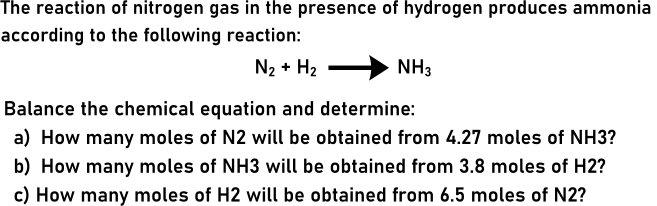
Transcribed Image Text:
The reaction of nitrogen gas in the presence of hydrogen produces ammonia according to the following reaction: N2 + H2 NH3 Balance the chemical equation and determine: a) How many moles of N2 will be obtained from 4.27 moles of NH3? b) How many moles of NH3 will be obtained from 3.8 moles of H2? c) How many moles of H2 will be obtained from 6.5 moles of N2? The reaction of nitrogen gas in the presence of hydrogen produces ammonia according to the following reaction: N2 + H2 NH3 Balance the chemical equation and determine: a) How many moles of N2 will be obtained from 4.27 moles of NH3? b) How many moles of NH3 will be obtained from 3.8 moles of H2? c) How many moles of H2 will be obtained from 6.5 moles of N2?
Expert Answer:
Answer rating: 100% (QA)
The Reachon of Nihogen gan inthe Prepence of Hydrogen Soluhion Produces amioni... View the full answer

Posted Date:
Students also viewed these chemistry questions
-
How many moles of Al2O3 will be produced when 23.9 g of H2O are reacted according to this chemical equation? 2AlCl3 + 3H2O() ( Al2O3 + 6HCl(g)
-
The solubility of nitrogen gas in rubber at 25C is 0.00156 kmol/m3 bar. When phase equilibrium is established, the density of nitrogen in a rubber piece placed in a nitrogen gas chamber at 800 kPa...
-
The solubility of nitrogen gas in rubber at 25C is 0.00156 kmol/m3bar. When phase equilibrium is established, the density of nitrogen in a rubber piece placed in a nitrogen gas chamber at 300 kPa is...
-
The accompanying table shows proportions of computer salespeople classified according to marital status and whether they left their jobs or stayed over a period of 1 year. a. What is the probability...
-
List the advantages and disadvantages of random sampling.
-
Assume that the company for which you are working is interested in implementing a comprehensive monitoring and control system regarding environmental performance. The company is convinced that...
-
a deontological philosopher, stated in An Exposition of The Fundamental Principles of Jurisprudence as the Science of Right that Judicial Punishment can never be administered merely as a means for...
-
Conway Manufacturing Company was started on January 1, 2014. The company was affected by the following events during its first year of operation. 1. Acquired $2,000 cash from the issue of common...
-
(a) Identify and explain the various ways through which government can intervene and assist upcoming Small entrepreneurs to grow and sustain their enterprises. (b) Explain the rationale behind SWOT...
-
This project is to help you apply concepts to real-world, real data analysis. In the real-world computers do most of the hard work for statisticians. This project will help you understand how an...
-
Find R=a+b for the vectors shown in Fig.1. Y R Z a a , X Vectors a and b make angles a and B with the horizontal (x) axis. Take a=3, b=4, a=30, B=609 Method 1, geometry of triangles. Use the formula...
-
Sheridan Service sells oil at a markup of 31% of the selling price. Sheridan paid $0.87 per litre of oil (a) What is the selling price per litre? (b) What is the rate of markup based on cost? (a) The...
-
Research shows consumers are leery about sharing their private information with researchers for fear they will be harmed and their confidential information will be _______. Group of answer choices...
-
Actual personal spending in 2007 chained dollars was $1,002.3 billion in 2015 and $1,026.2 billion in 2016. In current dollars, it was $1,115.6 billion in 2015 and $1,153.5 billion in 2016. . Source:...
-
12. In the following tasks, match the appropriate quality term with the example given. Choices are: Quality Management System (QMS), Quality Assurance (QA), or Quality Control (QC) a) Assessment of...
-
Both Medical Manufacturing and R&D Health Inc. have developed advanced hearing aid technologies. R&D's hearing aids sell at $4,000 per pair, whereas Medical Manufacturing sells its hearing aids at...
-
Describe what you see in the graph below. a. Explain what the variables are in the graph (X and Y axis - what they measure and what the units are.) b. Explain what the 4 lines are (purple, red,...
-
A non-charmed baryon has strangeness S = 2 and electric charge Q = 0. What are the possible values of its isospin I and of its third component I z ? What is it usually called if I = 1/2?
-
Compute parameter estimates for a regression model that is nonlinear in the parameters and explain how nonlinear least squares differs from linear least squares.
-
Explain the properties of the restricted least squares estimator. In particular, how do its bias and variance compare with those of the unrestricted, ordinary, least squares estimator?
-
Explain the concept of a control variable and the assumption necessary for a control variable to be effective.

Study smarter with the SolutionInn App

