The surface of copper gets tarnished by the formation of copper oxide. N gas was passed...
Fantastic news! We've Found the answer you've been seeking!
Question:
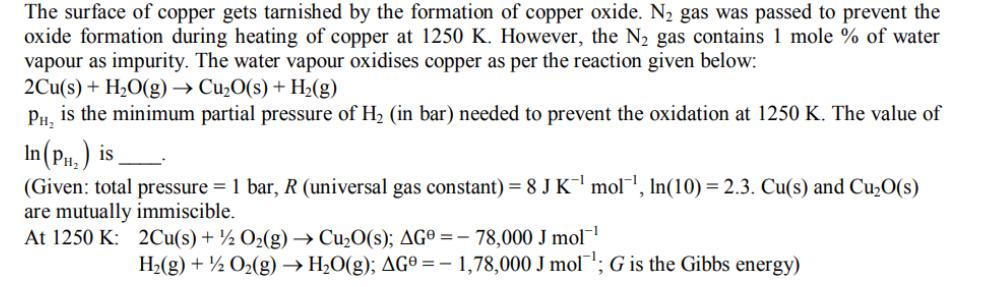
Transcribed Image Text:
The surface of copper gets tarnished by the formation of copper oxide. N gas was passed to prevent the oxide formation during heating of copper at 1250 K. However, the N gas contains 1 mole % of water vapour as impurity. The water vapour oxidises copper as per the reaction given below: 2Cu(s) + HO(g) CuO(s) + H(g) PH is the minimum partial pressure of H (in bar) needed to prevent the oxidation at 1250 K. The value of In (P) is (Given: total pressure = 1 bar, R (universal gas constant) = 8 J K mol, In(10) = 2.3. Cu(s) and CuO(s) are mutually immiscible. At 1250 K: 2Cu(s) + O(g) CuO(s); AG-78,000 J mol- H(g) + 1/2O(g) HO(g); AG = -1,78,000 J mol G is the Gibbs energy) The surface of copper gets tarnished by the formation of copper oxide. N gas was passed to prevent the oxide formation during heating of copper at 1250 K. However, the N gas contains 1 mole % of water vapour as impurity. The water vapour oxidises copper as per the reaction given below: 2Cu(s) + HO(g) CuO(s) + H(g) PH is the minimum partial pressure of H (in bar) needed to prevent the oxidation at 1250 K. The value of In (P) is (Given: total pressure = 1 bar, R (universal gas constant) = 8 J K mol, In(10) = 2.3. Cu(s) and CuO(s) are mutually immiscible. At 1250 K: 2Cu(s) + O(g) CuO(s); AG-78,000 J mol- H(g) + 1/2O(g) HO(g); AG = -1,78,000 J mol G is the Gibbs energy)
Expert Answer:
Related Book For 

Fundamentals Of Momentum Heat And Mass Transfer
ISBN: 9781118947463
6th Edition
Authors: James Welty, Gregory L. Rorrer, David G. Foster
Posted Date:
Students also viewed these chemistry questions
-
The vapour pressure of pure liquid A at 293 K is 68.8 kPa and that of pure liquid B is 82.1 kPa. These two compounds form ideal liquid and gaseous mixtures. Consider the equilibrium composition of a...
-
At 10 000 K the ionization reaction for Ar is: Ar Ar+ + e with equilibrium constant of K = 4.2 104. What should the pressure be for a mole concentration of argon ions (Ar+) of 10%?
-
At 10 000 K the two ionization reactions for N and Ar as have equilibrium constants of K1 = 4.2 104 and K2 = 6.3 104, respectively. If we start out with 1 kmol Ar and 0.5 kmol N2, what is the...
-
PWX Inc. has the following information for its years ended June 30: Required: Calculate the accounts receivable turnover and average collection period for 20X3 and 20X2. Comment on the trend. What...
-
Safe-Tee Glass Company manufactures three types of safety plate glass: large, medium, and small. All three products have high demand. Thus, Safe-Tee Glass is able to sell all the safety glass that it...
-
Using a webcam, a traffic analyst selected a random sample of 800 cars traveling on I-195 in Miami on a weekday morning. Among the 800 cars in the sample, 24% were white. The margin of error for this...
-
With reference to Exercise 3.25, suppose that three of the spark plugs are defective. In how many ways can 4 spark plugs be selected so that (a) one of the defective plugs is selected; (b) two of the...
-
After examining all the potential projects, you discover that there are many more projects this year with positive NPVs than in a normal year. What two problems might this extra large capital budget...
-
SOUTH DAKOTA CORPORATION SOUTH DAKOTA CORPORATION uses a job - order costing system with normal costing. At the start of the period, the company had the following balances: Raw Materials inventory $...
-
Alaska Airlines showed the following items in its balance sheet as of December 31, 2011, the end of the fiscal year ($ in millions): Inventories and supplies .............. $ 44.3 Prepaid expenses...
-
On December 31,2017, Jovial Company received two 1,000,000 notes receivable from customers in exchange for service rendered. On both notes, Interest is calculated on the outstanding principal balance...
-
The ACME Freight Co. has containerized a large quantity of 4-gigabyte memory chips that are to be shipped to a personal computer manufacturer in California. The shipment contains 1,000 boxes of...
-
A health maintenance organization (HMO) is currently treating 10 patients with a deadly bacterial infection. The best-known antibiotic treatment is being used in these cases, and this treatment is...
-
The joint density of the bivariate random variable \((X, Y)\) is given by \(f(x, y)=x y I_{[0,1]}(x) I_{[0,2]}(y)\). a. Find the joint cumulative distribution function of \((X, Y)\). Use it to find...
-
Amy is a partner in Xiowa Partners. Her basis in her partnership interest is $69,000. In a non-liquidating distribution, Amy received three properties, Property 1 was worth $20,000 and had a tax...
-
Let \(X\) be a random variable representing the minimum of the two numbers of dots that are facing up after a pair of fair dice is rolled. Define the appropriate probability density for \(X\). What...
-
You, a Captain, are a section chief in Military Personnel and 2d Lt Smith's supervisor. Lt Smith is a Force Support Officer. She has been on active duty for a year and in her present job for 10...
-
A genetically engineered strain of Escherichia coli (E. coli) is used to synthesize human insulin for people suffering from type I diabetes mellitus. In the following simplified reaction scheme,...
-
During the development of a 300-ft ship, it is desired to test a 10% scale model in a towing tank to determine the drag characteristics of the hull. Determine how the model is to be tested if the...
-
The mass transfer device shown in the figure at the top of the next column is used to carry out the controlled release of a vapor-phase pheromone drug used in pest control. The solid drug sublimes at...
-
In a velocity field where v = 400[(y/L) 2 e x + (x/L) 2 e y ] fps, determine the pressure gradient at the point (L, 2L). The y axis is vertical, the density is 64.4 Ib m /ft 3 and the flow may be...
-
Analysis of Canadian Manufacturing Workers Workhours. The time plot in Figure 18 . 22 describes the average annual number of weekly hours spent by Canadian manufacturing workers...
-
Souvenir Sales. The file SouvenirSales.csv contains monthly sales for a souvenir shop at a beach resort town in Queensland, Australia, between 1995 and 2001. [Source: Hyndman and Yang (2018).] Back...
-
Toys R US Revenues. Figure 18 . 23 is a time plot of the quarterly revenues of Toys R US between 1992 and 1995 (thanks to Chris Albright for suggesting the use of these data, which are available in...

Study smarter with the SolutionInn App



