Question: This is the chemical formula for hexanal: CH(CH) 4CHO. Calculate the mass percent of the carbon in hexanal.
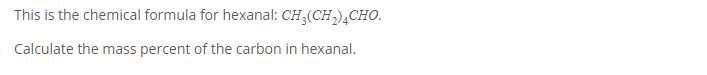
This is the chemical formula for hexanal: CH(CH) 4CHO. Calculate the mass percent of the carbon in hexanal.
Step by Step Solution
3.51 Rating (151 Votes )
There are 3 Steps involved in it
The mass percent of carbon in hexanal can be calculated using the following formula Mass ... View full answer

Get step-by-step solutions from verified subject matter experts


