Question: Three pipes A, B and C can fill a cistern in 10, 12, and 15 hours respectively, when working alone. If all the three
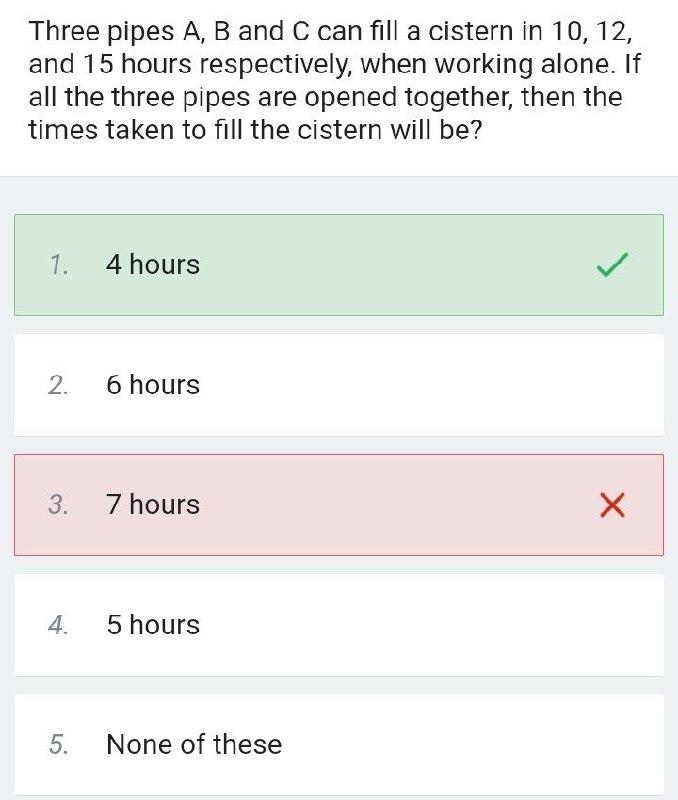
Three pipes A, B and C can fill a cistern in 10, 12, and 15 hours respectively, when working alone. If all the three pipes are opened together, then the times taken to fill the cistern will be? 1. 4 hours 2. 6 hours 3. 7 hours 4. 5 hours None of these 5.
Step by Step Solution
3.42 Rating (142 Votes )
There are 3 Steps involved in it
Solution 1 Since three pipes A B and C can fill a cistern in 10 12 and 1... View full answer

Get step-by-step solutions from verified subject matter experts


