2. Given the following biosynthesis scheme, answer the questions (a)-(c) below: OH O Me C E...
Fantastic news! We've Found the answer you've been seeking!
Question:
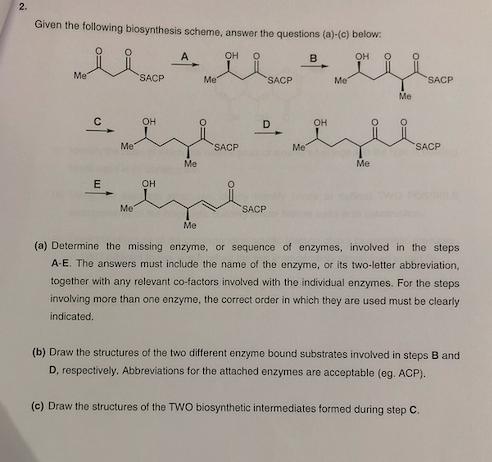
Transcribed Image Text:
2. Given the following biosynthesis scheme, answer the questions (a)-(c) below: OH O Me C E Me Me SACP OH OH A Me Me Me OH O SACP SACP D SACP Me B 4 OH Me Me Me SACP SACP (a) Determine the missing enzyme, or sequence of enzymes, involved in the steps A-E. The answers must include the name of the enzyme, or its two-letter abbreviation, together with any relevant co-factors involved with the individual enzymes. For the steps involving more than one enzyme, the correct order in which they are used must be clearly indicated. (b) Draw the structures of the two different enzyme bound substrates involved in steps B and i D, respectively. Abbreviations for the attached enzymes are acceptable (eg. ACP). (c) Draw the structures of the TWO biosynthetic intermediates formed during step C. 2. Given the following biosynthesis scheme, answer the questions (a)-(c) below: OH O Me C E Me Me SACP OH OH A Me Me Me OH O SACP SACP D SACP Me B 4 OH Me Me Me SACP SACP (a) Determine the missing enzyme, or sequence of enzymes, involved in the steps A-E. The answers must include the name of the enzyme, or its two-letter abbreviation, together with any relevant co-factors involved with the individual enzymes. For the steps involving more than one enzyme, the correct order in which they are used must be clearly indicated. (b) Draw the structures of the two different enzyme bound substrates involved in steps B and i D, respectively. Abbreviations for the attached enzymes are acceptable (eg. ACP). (c) Draw the structures of the TWO biosynthetic intermediates formed during step C.
Expert Answer:
Answer rating: 100% (QA)
Answer CS NG me B E A Me 4 23 step 11 are R c Me 2 acetyl CoA Acp traniacylare Bhy... View the full answer

Related Book For 

OM operations management
ISBN: 978-1285451374
5th edition
Authors: David Alan Collier, James R. Evans
Posted Date:
Students also viewed these chemistry questions
-
Prepare a cost of goods sold budget for Day Timer Publishers Inc. using the information in Practice Exercises 22-3B and 22-4B. Assume the estimated inventories on January 1, 2010, for finished goods...
-
Prepare a cost of goods sold budget for Soft Glow Candle Co. using the information in Practice Exercises 22-3A and 22-4A. Assume the estimated inventories on January 1, 2010, for finished goods and...
-
Prepare a cost of goods sold budget for Kenwood Manufacturing Inc. for the year ended December 31, 2013, from the following estimates. Inventories of production units: Direct materials purchased...
-
Summarize the extract given below? When Paul Farmer graduated from Duke University at 22, he was unsure whether he wanted to be an anthropologist or a doctor. So he went to Haiti. As a student, Paul...
-
A Starbucks barista wore a Wiccan medallion on a chain around her neck as part of her practice of the Wiccan religion. The store manager regularly made negative comments about the pendant, saying...
-
Which of the following is an example of a molecular marker? a. RFLP b. Microsatellite c. Single-nucleotide polymorphism d. All of the above are types of molecular markers.
-
Which account does a merchandiser use that a service company does not use? a. Cost of goods sold b. Inventory C. Sales revenue d. All of the above
-
Reitmans (Canada) Limited is a leading Canadian retailer that operates more than 900 stores under the Reitmans, Smart Set, RW & Co., Thyme Maternity, Penningtons, and Addition Elle banners. The...
-
Coke's most recent dividend was $1. Dividends are expected to grow by 15% for the next two years which would lead to dividends of $1.15 at time 1 and $1.32 at time 2. After that, dividends are...
-
The workload in many areas of bank operations has the characteristics of a nonuniform distribution with respect to time of day. For example, at Chase Manhattan Bank in New York, the number of...
-
A buffer solution is 0.356 M in H 2 SO 3 and 0.217 M in NAHSO 3 . If Ka1 for H 2 SO 3 is 1.7x10-2 , what is the pH of this buffer solution?
-
Lin Corporation has a single product, whose selling price is $200 and whose variable cost is 60% of sales price. The company's monthly fixed expenses are $60,000. Required: 1. Using the equation...
-
A gasoline tanker truck can hold 9000 gallons of gasoline. It isfilled with gasoline at a temperature of 68F (density is 1.32slug/ft3, dynamic viscosity is 0.61x10-6 lbs/ft2). It takes 2hours to fill...
-
Find the GCD and the LCM for each of the following groups of numbers using the intersection-of-sets method. a. 18 and 12 b. 28 and 36 c. 8, 40, and 64 d. 11 and 9 a. GCD(18,12)= LCM(18,12)= b....
-
organizational leadership is considering buying a competitor and has asked the software security team to develop a plan to ensure the competitors point of sale system complies with organization...
-
a) Write a program for a logic circuit given below with the help of gate-level modeling in Verilog. (2 marks) (2 marks) b) Write the logical expressions and a test bench of the given circuit diagram?
-
Using a Matlab plot the marks distribution of 62 students using histogram graph. The marks obtained by these students are [63 55 81 48 91 63 65 65 69 20 66 50 61 50 68 73 93 50 71 66 53 63 68 73 60...
-
What services are provided by the provincial and territorial governments?
-
Identify two competing organizations (for example, AT&T and Verizon, Taylor Made and Callaway golf club manufacturers, or Starbucks and McDonalds). Explain the differences in their missions,...
-
Explain how operations management activities affect the customer experiences described in the Museum of Science + Technology anecdote at the beginning of this chapter. What moments of truth would a...
-
Two experienced managers at Wilson Boat, Inc. are resisting the introduction of a computerized exponential smoothing system, claiming that their judgmental forecasts are much better than any computer...
-
You have been approached by promoters to give an opinion on the financial statements to be included in the prospectus of a proposed corporation to be named U-Park Corporation. U-Park will own and...
-
The following questions relate to the auditor's responsibility for reporting on inconsistency of application of accounting principles. Select the best response. a. Raider uses the last-in, first-out...
-
J.O. Cole, a sole proprietor, operates a trucking business. Cole also has assets and liabilities in connection with other activities. You are retained to audit the accounts of the J.O.C. Truck Lines...

Study smarter with the SolutionInn App


