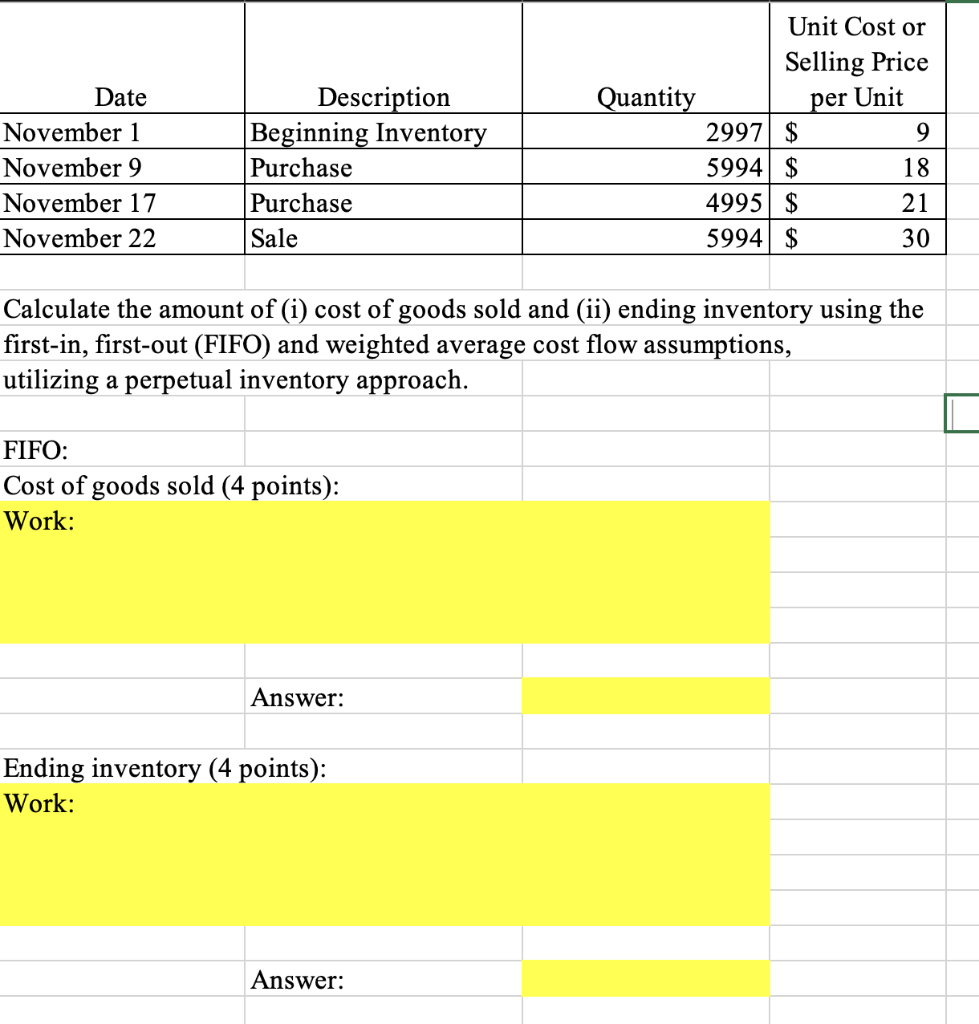
Unit Cost or Selling Price Description Beginning Inventory Purchase Purchase Date Quantity per Unit 2997 $...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
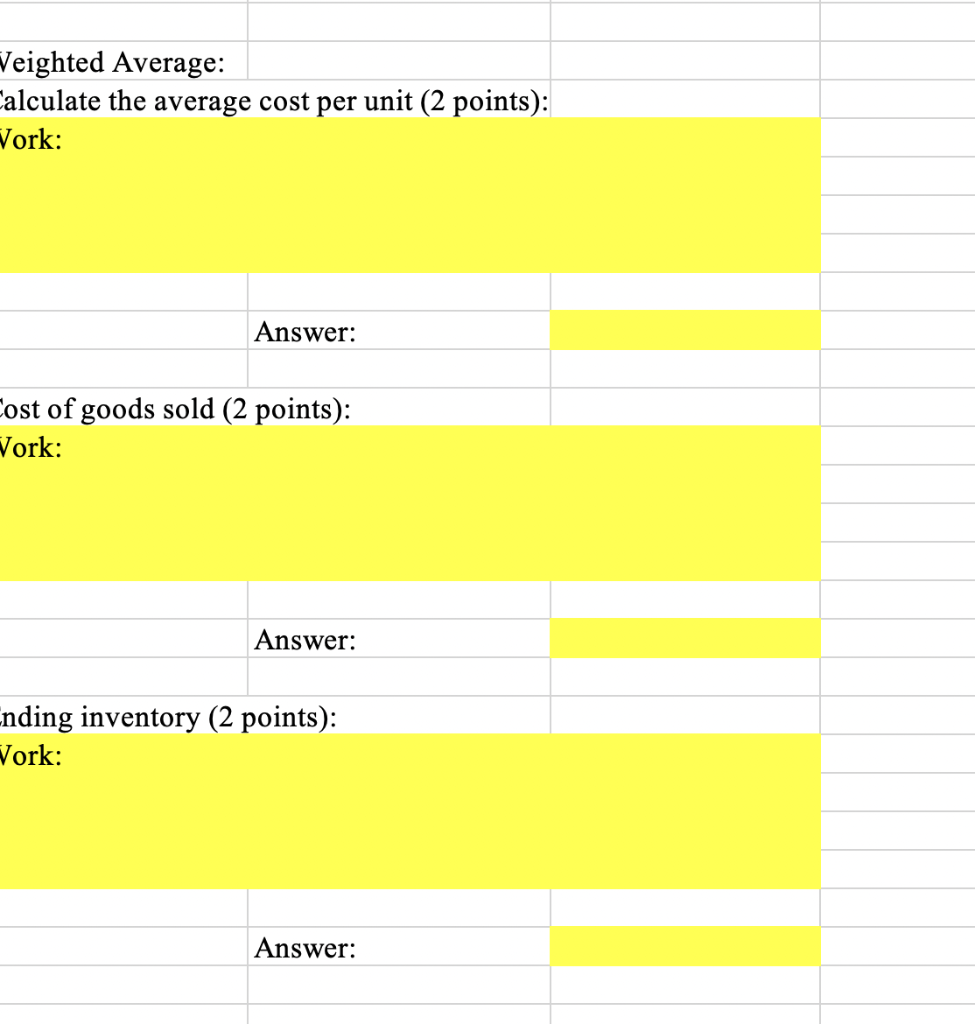
Unit Cost or Selling Price Description Beginning Inventory Purchase Purchase Date Quantity per Unit 2997 $ 5994 $ 4995 $ 5994 $ November 1 November 9 18 November 17 21 November 22 Sale 30 Calculate the amount of (i) cost of goods sold and (ii) ending inventory using the first-in, first-out (FIFO) and weighted average cost flow assumptions, utilizing a perpetual inventory approach. FIFO: Cost of goods sold (4 points): Work: Answer: Ending inventory (4 points): Work: Answer: Veighted Average: alculate the average cost per unit (2 points): Vork: Answer: ost of goods sold (2 points): Vork: Answer: nding inventory (2 points): Work: Answer: Unit Cost or Selling Price Description Beginning Inventory Purchase Purchase Date Quantity per Unit 2997 $ 5994 $ 4995 $ 5994 $ November 1 November 9 18 November 17 21 November 22 Sale 30 Calculate the amount of (i) cost of goods sold and (ii) ending inventory using the first-in, first-out (FIFO) and weighted average cost flow assumptions, utilizing a perpetual inventory approach. FIFO: Cost of goods sold (4 points): Work: Answer: Ending inventory (4 points): Work: Answer: Veighted Average: alculate the average cost per unit (2 points): Vork: Answer: ost of goods sold (2 points): Vork: Answer: nding inventory (2 points): Work: Answer: Unit Cost or Selling Price Description Beginning Inventory Purchase Purchase Date Quantity per Unit 2997 $ 5994 $ 4995 $ 5994 $ November 1 November 9 18 November 17 21 November 22 Sale 30 Calculate the amount of (i) cost of goods sold and (ii) ending inventory using the first-in, first-out (FIFO) and weighted average cost flow assumptions, utilizing a perpetual inventory approach. FIFO: Cost of goods sold (4 points): Work: Answer: Ending inventory (4 points): Work: Answer: Veighted Average: alculate the average cost per unit (2 points): Vork: Answer: ost of goods sold (2 points): Vork: Answer: nding inventory (2 points): Work: Answer: Unit Cost or Selling Price Description Beginning Inventory Purchase Purchase Date Quantity per Unit 2997 $ 5994 $ 4995 $ 5994 $ November 1 November 9 18 November 17 21 November 22 Sale 30 Calculate the amount of (i) cost of goods sold and (ii) ending inventory using the first-in, first-out (FIFO) and weighted average cost flow assumptions, utilizing a perpetual inventory approach. FIFO: Cost of goods sold (4 points): Work: Answer: Ending inventory (4 points): Work: Answer: Veighted Average: alculate the average cost per unit (2 points): Vork: Answer: ost of goods sold (2 points): Vork: Answer: nding inventory (2 points): Work: Answer: Unit Cost or Selling Price Description Beginning Inventory Purchase Purchase Date Quantity per Unit 2997 $ 5994 $ 4995 $ 5994 $ November 1 November 9 18 November 17 21 November 22 Sale 30 Calculate the amount of (i) cost of goods sold and (ii) ending inventory using the first-in, first-out (FIFO) and weighted average cost flow assumptions, utilizing a perpetual inventory approach. FIFO: Cost of goods sold (4 points): Work: Answer: Ending inventory (4 points): Work: Answer: Veighted Average: alculate the average cost per unit (2 points): Vork: Answer: ost of goods sold (2 points): Vork: Answer: nding inventory (2 points): Work: Answer: Unit Cost or Selling Price Description Beginning Inventory Purchase Purchase Date Quantity per Unit 2997 $ 5994 $ 4995 $ 5994 $ November 1 November 9 18 November 17 21 November 22 Sale 30 Calculate the amount of (i) cost of goods sold and (ii) ending inventory using the first-in, first-out (FIFO) and weighted average cost flow assumptions, utilizing a perpetual inventory approach. FIFO: Cost of goods sold (4 points): Work: Answer: Ending inventory (4 points): Work: Answer: Veighted Average: alculate the average cost per unit (2 points): Vork: Answer: ost of goods sold (2 points): Vork: Answer: nding inventory (2 points): Work: Answer:
Expert Answer:
Answer rating: 100% (QA)
Answer Page Cost of Ending inventos SOLN Finst in Finst out Cost o... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Calculate the amount of work input a refrigerator needs to make ice cubes out of a tray of 0.25 kg liquid water at 10oC. Assume the refrigerator has = 3.5 and a motor-compressor of 750 W. How much...
-
Calculate the amount of work input a refrigerator needs to make ice cubes out of a tray of 0.5 lbm liquid water at 50 F. Assume the refrigerator has = 3.5 and a motor-compressor of 750 W. How much...
-
Calculate the amount of work input a refrigerator needs to make ice cubes out of a tray of 0.25 kg liquid water at 10oC. Assume the refrigerator works in a Carnot cycle between 8oC and 35oC with a...
-
A pack of iron bolts is such that the difference in masses or weights of successive sizes is the same bolt being of mass 13.5 grams and the largest is 94.5 grams. If the total mass of the complete...
-
Hoop It Up has two classes of stock authorized: 7%, $20 par preferred and $1 par value common. The following transactions affect stockholders equity during 2015, its first year of operations:...
-
Exhibit 14.12 presents market and profit data for three companies. If Company 3 has nonoperating assets valued at $50 million, what are the company's appropriate enterprise-value-to-EBITDA and...
-
The following information is taken from the annual report of The Coca-Cola Company: Using this information, calculate the accounts payable turnover ratio and the days payable period for 2015 and...
-
Jamie Lee Jackson, age 26, is in her last semester of college and is anxiously waiting for graduation day that is just around the corner! She still works part-time as a bakery clerk, has been...
-
Question 3 . a ) What is heat capacity? B ) What is thermal expansion? C ) What is thermal conductivity?
-
A large company is planning to downsize. A consultant suggests the following: 1) It makes sense to focus on one of your unionized plants. We suggest that you reduce the 300 employees at that plant to...
-
Toplob Inc. ("Toplob") provides employment consulting services. These services range from maintaining payroll records to taxation services, as well as general business advisory and consulting to new...
-
A tank contains \(400 \mathrm{~kg}\) of a fluid. If the volume of the tank is \(2.5 \mathrm{~m}^{3}\), then what is the density of the fluid and what is the specific gravity?
-
What is the difference between steady and uniform states?
-
A spherical ball of mass \(10 \mathrm{~kg}\) is dropped from a height of \(15 \mathrm{~m}\). What is its potential energy at the time of release? Consider the potential energy of the ball as...
-
'All spontaneous processes are irreversible'. Explain with a common example.
-
Customers arrive an average of 10 per hour and an average of 16 customers can be served in an hour. Assuming this is an M/M/1 model, what is the average amount of time a customer spends in the system?
-
In a classic ecological paper by Crouse et al., the authors assume that adult loggerhead turtles have (approximately) an 80% chance of surviving from year to year. Let X be the number of years that a...
-
Currently, there are five concepts of food stands, including: hot dogs, soft pretzels, turkey legs, sandwich wraps, and funnel cakes. This approach will double the existing number of food stands...
-
Identify each of the following as an adverse selection or a moral hazard problem. (a) A person with car insurance fails to lock his car doors when he shops at a mall. (b) A person with a family...
-
1. Describe when asymmetric information exists. 2. Explain under what condition asymmetric information leads to market failure. 3. Name the asymmetric information problem that exists prior to an...
-
With respect to the interest rate, (a) What is the liquidity effect? (b) What is the price-level effect? (c) What is the expectations effect?
-
A distillation column operating at total reflux is separating acetone and ethanol at \(1.0 \mathrm{~atm}\). The height of packing is \(2.0 \mathrm{~m}\). The column has a partial reboiler and a total...
-
We wish to strip \(\mathrm{SO}_{2}\) from water using pure air at \(20.0^{\circ} \mathrm{C}\). Outlet water contains \(0.0060 \mathrm{~mol} \% \mathrm{SO}_{2}\), and inlet water contains \(0.112...
-
If 1-in. metal Raschig rings are used instead of 2-in. rings in Example \(16-2\) : Example 16-2 Example 4-3 Example 16-1 a. Recalculate the flooding velocity and the required diameter. b. Recalculate...

Study smarter with the SolutionInn App


