Question: Useful I data: Y = C/ C 1.4 Gas Molar mass- M-, kg/k mol Air 28.97 32 1.4 en Butane (C,H10) Nitrogen (N2) Methane
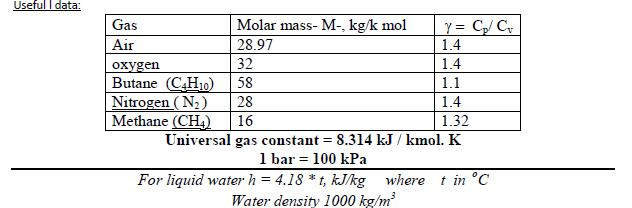

Useful I data: Y = C/ C 1.4 Gas Molar mass- M-, kg/k mol Air 28.97 32 1.4 en Butane (C,H10) Nitrogen (N2) Methane (CH) 58 1.1 28 1.4 16 1.32 Universal gas constant = 8.314 kJ / kmol. K 1 bar = 100 kPa For liquid water h = 4.18 *t, kJ/kg where t in C Water density 1000 kg/m 10- Methane (CH,) gas enters a horizontal, well-insulated nozzle operating at steady state at 80C and a velocity of 10 m/s. Assuming ideal gas behavior for the methane, plot the temperature of the gas exiting the nozzle, in C, versus the exit velocity ranging from 500 to 600 m/s.
Step by Step Solution
3.42 Rating (152 Votes )
There are 3 Steps involved in it
To solve this problem well apply the principles of fluid dynamics and thermodynamics specifically focusing on energy conservation Bernoullis equation ... View full answer

Get step-by-step solutions from verified subject matter experts


