Using the figure on the following page on how to achieve an octet for cations and...
Fantastic news! We've Found the answer you've been seeking!
Question:

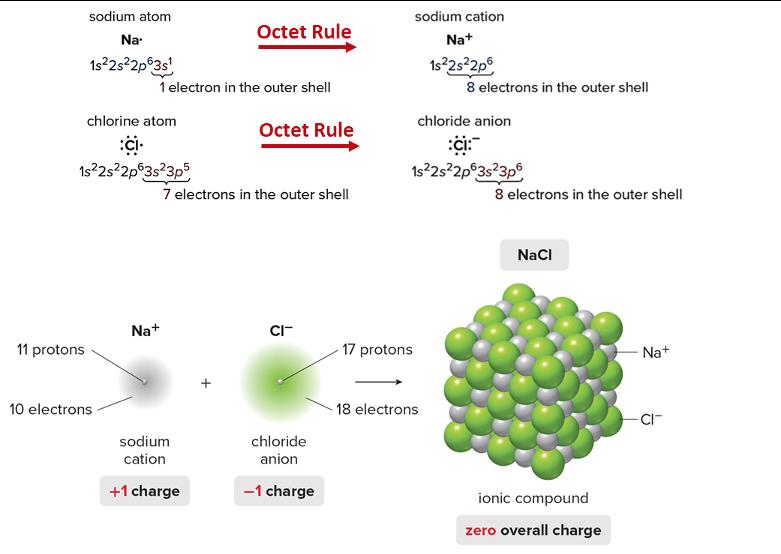
Transcribed Image Text:
Using the figure on the following page on how to achieve an octet for cations and anions, please draw a Lewis dot structure for each of the following elements. Also, identify what net charge each element would adopt to achieve the valence electron configuration of a noble gas (achieve a stable octet). ***See Lithium below as an example. Li Lit where it has 1+ net charge a. Example: Li Lewis Structure: b. F Lewis structure: c. Mg Lewis structure: d. Cl Lewis Structure: e. K Lewis structure: f. S Lewis structure: g. Ca sodium atom Na- 1s²2s²2p63s¹ 11 protons chlorine atom :CI 1s²2s²2p63s²3p5 10 electrons 1 electron in the outer shell Na+ Octet Rule sodium cation +1 charge Octet Rule 7 electrons in the outer shell CI- chloride anion -1 charge sodium cation Na+ 1s²2s²2p6 17 protons chloride anion :ci: 1s²2s²2p63s²3p6 8 electrons in the outer shell 18 electrons 8 electrons in the outer shell NaCl ionic compound zero overall charge Na+ -CI- Using the figure on the following page on how to achieve an octet for cations and anions, please draw a Lewis dot structure for each of the following elements. Also, identify what net charge each element would adopt to achieve the valence electron configuration of a noble gas (achieve a stable octet). ***See Lithium below as an example. Li Lit where it has 1+ net charge a. Example: Li Lewis Structure: b. F Lewis structure: c. Mg Lewis structure: d. Cl Lewis Structure: e. K Lewis structure: f. S Lewis structure: g. Ca sodium atom Na- 1s²2s²2p63s¹ 11 protons chlorine atom :CI 1s²2s²2p63s²3p5 10 electrons 1 electron in the outer shell Na+ Octet Rule sodium cation +1 charge Octet Rule 7 electrons in the outer shell CI- chloride anion -1 charge sodium cation Na+ 1s²2s²2p6 17 protons chloride anion :ci: 1s²2s²2p63s²3p6 8 electrons in the outer shell 18 electrons 8 electrons in the outer shell NaCl ionic compound zero overall charge Na+ -CI-
Expert Answer:
Answer rating: 100% (QA)
Solution z atomic number a Li15251 Lewis 23 5 b F H0 23 pps Lewis 29 ... View the full answer

Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Posted Date:
Students also viewed these chemistry questions
-
Draw a Lewis dot structure for each of the following compounds: a. CH 3 CH 2 OH b. CH 3 CN
-
Draw a Lewis dot structure for each of the following atoms: (a) Carbon (b) Oxygen (c) Fluorine (d) Hydrogen (e) Bromine (f) Sulfur (g) Chlorine (h) Iodine
-
The figure on the following page shows a unit circle, which will be used to illustrate a number of the subtasks. (A) For the angle a, sin(a)= -13/20 and - < < -1/2-. Mark a in a hot circle. (Use a...
-
Suppose that \(\mathrm{x}\) is a linked-list Node. What is the effect of the following code fragment? \(t\). next \(=x\). next; \(x \cdot\) next \(=t ;\)
-
Van Dyck Company has Determine that the variable overhead rate is $3.90 per direct labor hour in the Fabrication Department. The normal production capacity for the Fabrication Department is 14,000...
-
Assume 120 V is applied to the primary of a transformer with a VA rating of 10 VA. The output voltage is 12.6 V. What is the smallest resistor that can be connected across the secondary?
-
Describe the shifts in UK share ownership over the past 60 years. Suggest some reasons for the changes.
-
January budgeted selling and administrative expenses for the retail shoe store that Kathy Sibley plans to open on January 1, 2011, are as follows: sales commissions, $25,000; rent, $16,000;...
-
At December 31, 2023, Cord Company's plant asset and accumulated depreciation and amortization accounts had balances as follows: Category Land Accumulated Depreciation and Amortization $ Plant Asset...
-
Jack Tasker opened his Auto Repair Shop in November 2023. The balance sheet at November 30, 2023, prepared by an inexperienced part-time bookkeeper, is shown below. Required Prepare a correct balance...
-
Review the following: Slide show - 10 Cool CRM DevelopmentsLinks to an external site. Video - Brunello and SalesforceLinks to an external site. Discuss: 3 things you learned Locate an additional...
-
The following data show the ages of 50 patients who had sports-related surgeries. Construct a frequency distribution for the data using five classes and summarize the results. SEARC REESE 7253 28 25...
-
Why should the receiving department be segregated from inventory control and, if possible, from shipping?
-
What control objective is served by management implementing written procedures for all additions, disposals, and retirements? a. Procedures for operating, using, and physically moving plant assets...
-
What control objective is served by management's establishing policies for selecting and approving investment transactions? a. Sources of capital funds should be authorized in accordance with...
-
Why should policies assure that goods and services be purchased oiJy from approved vendors?
-
A company is setting its direct materials and direct labor standards for its leading product. Direct materials cost from the supplier are $5.00 per square foot, net of purchase discount. Freight-in...
-
Phosgene, COCl2, is a toxic gas used in the manufacture of urethane plastics. The gas dissociates at high temperature. At 400oC, the equilibrium constant Kc is 8.05 104. Find the percentage of...
-
Resolution with HPLC Teaching Assistant, an Excel spreadsheet that simulates high performance liquid chromatography separations. Download the Excel file from SaplingPlus, macmillanlearning.com, or...
-
What is the purpose of the WO3 WO 3 and Cu Cu in Figure 27-8? Figure 27-8 Leee Oxygen WO oxidation catalyst Cu reduction catalyst and O scavenger 7. Encapsulated sample Ceramic crucible Helium...
-
Select the fluorescent derivatization reagent from 1 to 3 for each of the following applications from the literature. a. Quantitation of thiols such as glutathione, cysteine, homocysteine, and y y...
-
In 2002, following its adoption of a new accounting standard issued by the FASB for the impairment of goodwill accounting, AMR Corporation, the parent company of American Airlines, conducted an...
-
Discuss the concepts of book value, intrinsic value, and market capitalization. Using a financial website such as YahooFinance or MSNMoney, identify the market capitalization and book value of The...
-
The chart below summarizes the U.S. exchange traded companies that issued corrections to their financial statements during the years 2001 through 2014. The data was compiled by Audit Analytics and...

Study smarter with the SolutionInn App


