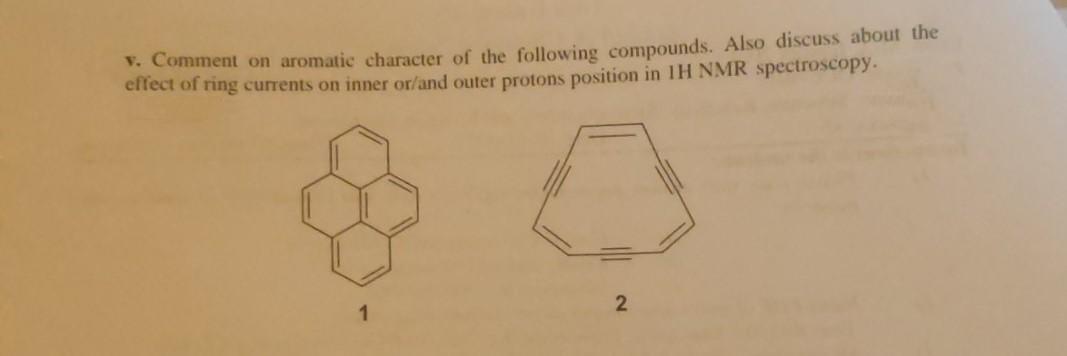
V. Comment on aromatic character of the following compounds. Also discuss about the elfect of ring...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
V. Comment on aromatic character of the following compounds. Also discuss about the elfect of ring currents on inner or/and outer protons position in 1H NMR spectroscopy. 2 V. Comment on aromatic character of the following compounds. Also discuss about the elfect of ring currents on inner or/and outer protons position in 1H NMR spectroscopy. 2
Expert Answer:
Answer rating: 100% (QA)
1 is pyrene it is aromatic in nature because its have 14 delocalized electrons insid... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Discuss how you can determine which of the following compounds might be alkanes, cycloalkanes, alkenes, or alkynes, without drawing their formulas: (a) C6H12. (b) C4H6. (c) C5H12. (d) C7H14, (e) C3H4.
-
Order the 1H NMR signals of the following compounds by chemical-shift position (lowest to highest). Which one is the most upheld? The most downfield? (a) H3C-CH3 (b) H2C==CH2 (c) H3C-O-CH3 (d) (e)...
-
Each of the following compounds has a single 1H NMR peak. Approximately where would you expect each compound toabsorb? (c) (b) (a) "H (d) CH2CI2 (e) O ( N-CH C-C
-
Let y vary directly with x. Complete the following. Find y when x = 1, if y = when x = 3.
-
You're a marketing manager for Stanton, one of the premier suppliers of DJ equipment (turntables, amplifiers, speakers, mixers, and related accessories). Your company's latest creation, the...
-
For this exercise, you will electronically exchange a document with one of your classmates. With the Track Changes feature turned on, you will review each others documents, make comments or ask...
-
Assuming the same data as given in problem 9, was the well in each case profitable? Discuss your answer. Problem 9:- Property cost (acquisition cost). Drilling cost (one well). Estimated completion...
-
Calculating acquisition costs of long-lived assets. Outback Steakhouse opened a new restaurant on the site of an existing building. It paid the owner $260,000 for the land and building, of which it...
-
2. 2. A 20 kg child climbs to the top of a slide that is 3 m above the ground level. She starts from rest and slides down the incline. a. Define and model the energy of the system with Energy Bar...
-
RZNY, LP., is a calendar-year, cash-basis limited partnership. Marc Liu, an individual, owns a 55% interest in RZNY as a general partner. During Year 1, RZNY had the following results: 1. Sales -...
-
The total cost after tax to repair Hannah's fridge is represented by 0.06(45h)+ 45h, where h represents the number of hours it takes to repair the fridge. What part of the expression represents the...
-
Discuss which of the theories from HRTM 7708 you think is most consistent with your philosophy on managing people in the hospitality & tourism industry. Why? Would your response have been different...
-
A car dealership claims to give customers a median trade-in offer of at least $6000. A random sample of 103 transactions revealed that the trade-in offer for 60 automobiles was less than $6000 and...
-
1. why are digital payments disruptive to traditional banking? 2.Will we value an ICO and an STO differently? Why? 3. Do you think the abnormal price pattern is sustainable in the long run? Why or...
-
Calculate RT for the circuit below. R1 = 18 kQQ, R2 = 11 kQQ, and R3 = 19 kQ R2 Vs Answer:
-
write a class that satisfies the PersonDriver: The class will contain the following: An abstract Person class that contains one abstract method "public abstract String doesForALiving()" An interface...
-
Whenconsideringagloballogisticspartner,whatisanimportantcharacteristicthatshouldbeconsideredwhendevelopingarelationship?Explain.
-
What is a manufacturing system?
-
The combustion of 20.63 mg of compound Y, which contains only C, H, and O, with excess oxygen gave 57.94 mg of CO2 and 11.85 mg of H2O. (a) Calculate how many milligrams of C, H, and O were present...
-
How many different tripeptides can be formed by lysine and alanine?
-
Which of the following does not represent a Lewis acid-base reaction? (a) H2O + H+ H3O+ (b) NH3 + BF3 H3NBF3 (c) PF3 + F2 PF5 (d) Al(OH)3 + OH2 Al(OH)4-
-
The latent heat of vaporization per unit mass of a pure substance at a given temperature, \(\lambda\), is defined as the difference in enthalpy between the saturated vapor and saturated liquid at the...
-
It is desired to dehumidify \(1.2 \mathrm{~m}^{3} / \mathrm{s}\) of air, available at \(311 \mathrm{~K}\) with a wet-bulb temperature of \(303 \mathrm{~K}\), to a wet-bulb temperature of \(288...
-
Modify the Mathcad program developed in Problem 3.16 to estimate the minimum gas flow rate in strippers so that it can be used to estimate the minimum air flow required for water cooling. Test your...

Study smarter with the SolutionInn App


