Question: We have a distillation column with a partial condenser and a total reboiler separating a feed of 200 kmol/h. The feed is 40 mol
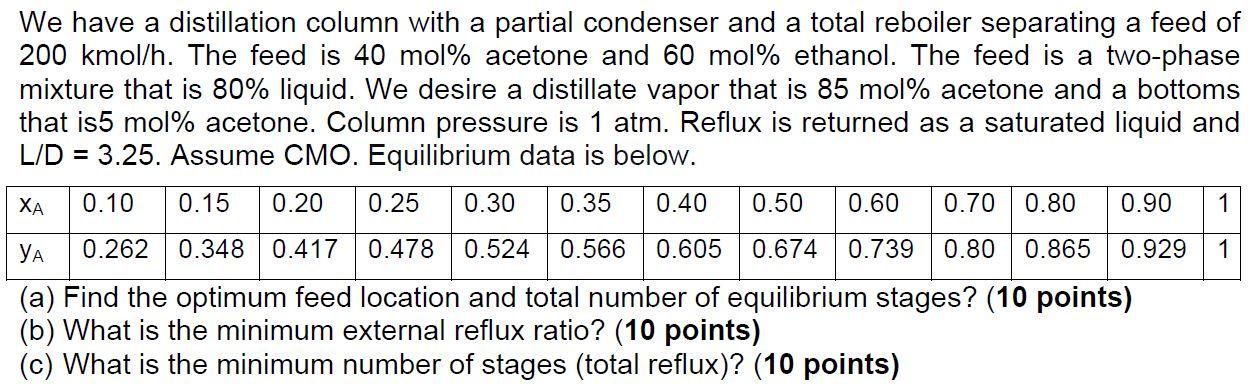
We have a distillation column with a partial condenser and a total reboiler separating a feed of 200 kmol/h. The feed is 40 mol % acetone and 60 mol % ethanol. The feed is a two-phase mixture that is 80% liquid. We desire a distillate vapor that is 85 mol % acetone and a bottoms that is5 mol% acetone. Column pressure is 1 atm. Reflux is returned as a saturated liquid and L/D = 3.25. Assume CMO. Equilibrium data is below. XA 0.10 0.15 0.20 0.25 0.30 0.35 0.40 0.50 0.60 0.70 0.80 0.90 1 0.262 0.348 0.417 0.478 0.524 0.566 0.605 0.674 0.739 0.80 0.865 0.929 1 (a) Find the optimum feed location and total number of equilibrium stages? (10 points) (b) What is the minimum external reflux ratio? (10 points) (c) What is the minimum number stages (total reflux)? (10 points)
Step by Step Solution
3.54 Rating (157 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


