Question: Weight Figure 1 (below) depicts a weighting function, which is sometimes described as an inverted S-curve. 1.0 b. C. 0.9 0.8 0.7 0.6- 0.5
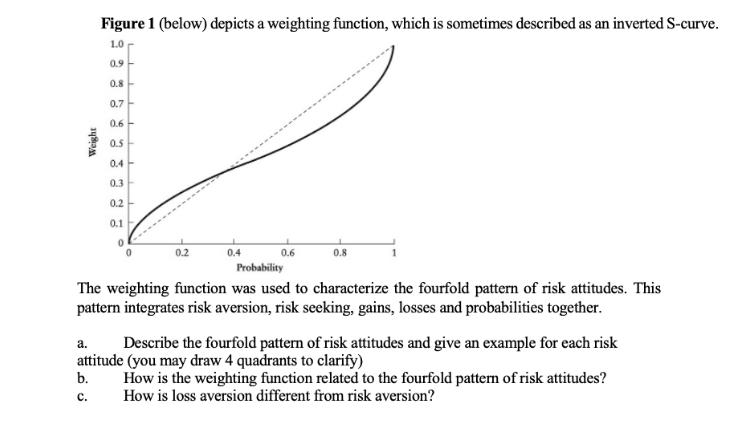
Weight Figure 1 (below) depicts a weighting function, which is sometimes described as an inverted S-curve. 1.0 b. C. 0.9 0.8 0.7 0.6- 0.5 L 0.4 0.3 0.2 0.1 + L 0.2 0.4 0.6 0.8 Probability The weighting function was used to characterize the fourfold pattern of risk attitudes. This pattern integrates risk aversion, risk seeking, gains, losses and probabilities together. a. Describe the fourfold pattern of risk attitudes and give an example for each risk attitude (you may draw 4 quadrants to clarify) How is the weighting function related to the fourfold pattern of risk attitudes? How is loss aversion different from risk aversion?
Step by Step Solution
3.38 Rating (154 Votes )
There are 3 Steps involved in it
a The fourfold pattern of risk attitudes also known as the fourfold pattern of risk preferences is a conceptual framework that categorizes individuals based on their attitudes towards risk and potenti... View full answer

Get step-by-step solutions from verified subject matter experts


