what is the net profit after dropping all the customers? Corley Communications performed a customer profitability analysis
Fantastic news! We've Found the answer you've been seeking!
Question:
what is the net profit after dropping all the customers?
Transcribed Image Text:
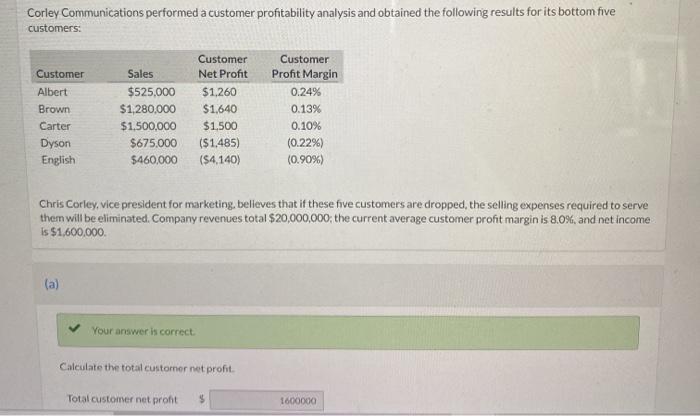
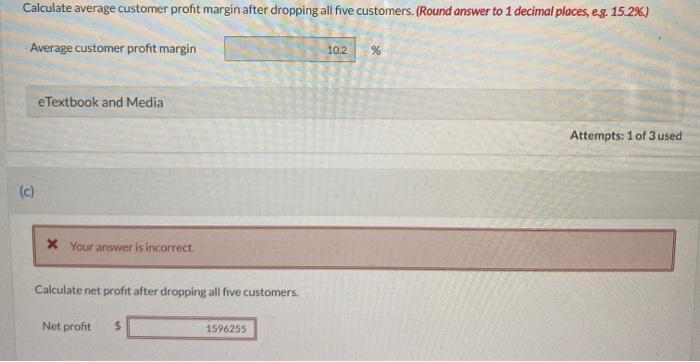
Corley Communications performed a customer profitability analysis and obtained the following results for its bottom five customers: Customer Albert Brown Carter Dyson English (a) Customer Net Profit $1,260 Sales $525,000 $1,280,000 $1,640 $1,500,000 $1,500 $675.000 ($1,485) $460,000 ($4,140) Chris Corley, vice president for marketing, believes that if these five customers are dropped, the selling expenses required to serve them will be eliminated. Company revenues total $20,000,000; the current average customer profit margin is 8.0%, and net income is $1,600,000. Your answer is correct. Customer Profit Margin 0.24% 0.13% 0.10% (0.22%) (0.90%) Calculate the total customer net profit. Total customer net profit 1600000 Corley Communications performed a customer profitability analysis and obtained the following results for its bottom five customers: Customer Albert Brown Carter Dyson English (a) Customer Net Profit $1,260 Sales $525,000 $1,280,000 $1,640 $1,500,000 $1,500 $675.000 ($1,485) $460,000 ($4,140) Chris Corley, vice president for marketing, believes that if these five customers are dropped, the selling expenses required to serve them will be eliminated. Company revenues total $20,000,000; the current average customer profit margin is 8.0%, and net income is $1,600,000. Your answer is correct. Customer Profit Margin 0.24% 0.13% 0.10% (0.22%) (0.90%) Calculate the total customer net profit. Total customer net profit 1600000
Expert Answer:
Answer rating: 100% (QA)
Ca Total customer net profit Total Revenue profit margin i... View the full answer

Related Book For 

Posted Date:
Students also viewed these general management questions
-
What is the net impact on Dos Santos Companys 2011 net income as a result of this hedge of a forecasted foreign currency transaction? a. $0. b. $400 decrease in net income. c. $1,000 decrease in net...
-
What is the net impact on Black Lion Company's Year 2 net income as a result of this hedge of a forecasted foreign currency purchase? Assume that the raw materials are consumed and become a part of...
-
What is the net impact on Black Lion Company's Year 1 net income as a result of this hedge of a forecasted foreign currency purchase? a. $0. b. A $200 increase in net income. c. A $300 decrease in...
-
1. Calculate the budgeted nights booked: Maximum capacity (30 rooms) * Number of days per year (365) * Expected occupancy rate (80%) = 8760 nights. 2. Calculate the tariff revenues: Budgeted nights...
-
Bosco Company adopted the dollar-value LIFO retail method at the beginning of 2011. Information for 2011 and 2012 is as follows, with certain data intentionally omitted: Required: Determine the...
-
The work done by one mole of a monatomic ideal gas ( = 5/3) in expanding adiabatically is 825 J. The initial temperature and volume of the gas are 393 K and 0.100 m3. Obtain (a) The final temperature...
-
The following contingency table presents observed frequencies. a. Compute the expected frequencies. b. One of the observed frequencies is less than 5. Is it appropriate to perform a test of...
-
Recall the Innis Investments problem. Letting S = units purchased in the stock fund M = units purchased in the money market fund Leads to the following formulation: Min8S + 3M s.t. 50S + 100M ...
-
The Houston Corporation manufactures filing cabinets in two operations: machining and finishing. It provides the following information: (Click the icon to view the department information.) Each...
-
Cullumber Co. uses a perpetual inventory system and both an accounts receivable and an accounts payable subsidiary ledger. Balances related to both the general ledger and the subsidiary ledger for...
-
a ) Over 4 3 , 0 0 0 students graduate from NZ universities each year. Graduates work for 4 0 years on average until retirement. How many graduates will be employed in 2 0 5 0 ? Explain the...
-
The table below reports real GDP per capita for Germany and the United States for 2008 and 2018. Country Germany the United States Per capita real GDP 2008 Per capita real GDP 2018 $42,102 $49,319...
-
Gravel is being dumped from a conveyor belt at a rate of 20 cubic feet per minute. It forms a pile in the shape of a right circular cone whose base diameter and height are always the same. How fast...
-
Do you think the guidelines in the voluntary standard can be helpful?
-
Which is more important: Resources or industry? In the big picture of things, there are two opposing strategic views on the most important contributor to above average returns. Usually companies are...
-
A particle is moving along the curve y = 52x+5. As the particle passes through the point (2, 15), its x- coordinate increases at a rate of 3 units per second. Find the rate of change of the distance...
-
In the context of technological advancements, outsourcing, and offshoring, some informed observers note that technology already has eliminated much of the work done by Question 5 options: top level...
-
What are the two methods used to translate financial statements and how does the functional currency play a role in determining which method is used?
-
Mighty Lights produces decorative lamps in several styles and finishes. The company uses a job order costing system to accumulate product costs. Because much of the production process is automated,...
-
A company that wants to "empower athletes everywhere," started out to produce a superior T-shirt that would help regulate an athlete's body temperature during workouts. The company has expanded its...
-
Paula Boothe, president of the Armange Corporation, has mandated a minimum 10% return on investment for any project undertaken by the company. Given the company's decentralization, Paula leaves all...
-
The following experimental data are available for \(\mathrm{CO}_{2}\) gas at \(92^{\circ} \mathrm{C}\) : PR 1 2 3 4 6 8 10 0.856 0.583 0.535 0.620 0.800 0.975 1.160 Find the fugacity of CO2 at 100...
-
What do you mean by extent of reaction? Derive an expression of the relationship between mole fraction of the component and the extent of reaction.
-
Give an informatory note on equilibrium constant of the chemical reaction.

Study smarter with the SolutionInn App


