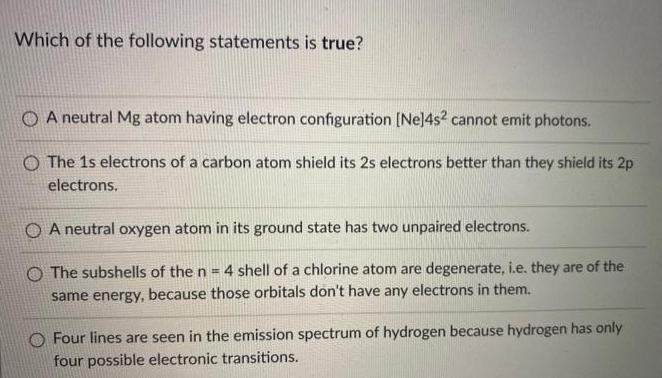
Which of the following statements is true? O A neutral Mg atom having electron configuration [Ne]4s?...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Which of the following statements is true? O A neutral Mg atom having electron configuration [Ne]4s? cannot emit photons. O The 1s electrons of a carbon atom shield its 2s electrons better than they shield its 2p electrons. O A neutral oxygen atom in its ground state has two unpaired electrons. O The subshells of the n = 4 shell of a chlorine atom are degenerate, i.e. they are of the same energy, because those orbitals don't have any electrons in them. O Four lines are seen in the emission spectrum of hydrogen because hydrogen has only four possible electronic transitions. Which of the following statements is true? O A neutral Mg atom having electron configuration [Ne]4s? cannot emit photons. O The 1s electrons of a carbon atom shield its 2s electrons better than they shield its 2p electrons. O A neutral oxygen atom in its ground state has two unpaired electrons. O The subshells of the n = 4 shell of a chlorine atom are degenerate, i.e. they are of the same energy, because those orbitals don't have any electrons in them. O Four lines are seen in the emission spectrum of hydrogen because hydrogen has only four possible electronic transitions.
Expert Answer:

Related Book For 

Accounting concepts and applications
ISBN: 978-0538745482
11th Edition
Authors: Albrecht Stice, Stice Swain
Posted Date:
Students also viewed these chemistry questions
-
which of the following statements is true select one a. an employer cannot be held liable for a tort that an employee deliberately committed b. vicarious liability supports tort law's compensation...
-
Which statement is true regarding recapture of listed property under 280F? The year following the year of recapture, use the straight-line (SL) method in the general depreciation system (GDS), as...
-
Which of the following statements is true about health savings accounts (HSAs)? Select one: a. Taxpayers may take tax and penalty free distributions from HSAs to purchase automobiles after age 65. b....
-
Cummings Corp. has three business segmentsthe Plumbing Parts division, Small Tools division, and Design Printing division. Carol Jenkins (Cummings' CFO) and William Samuels (head of Production) would...
-
A short-run total cost curve is given by the equation STC(Q) = 1000 + 50Q2. Derive expressions for, and then sketch, the corresponding short-run average cost, average variable cost, and average fixed...
-
Find the g.c.f. and l.c.m. of the sets of numbers in Problems 2934. {85, 100, 240}
-
Given the feed streams and the parameters of the process units as shown in Figure 7.38, complete the simulation flowsheet for ASPEN PLUS and show the calculation sequence (i.e., complete the...
-
Two-dimensional, steady-state conduction occurs in a hollow cylindrical solid of thermal conductivity k = 16 W/m K, outer radius ro = 1 m, and overall length 2zo = 5 m, where the origin of the...
-
You work for a large healthcare delivery system that is rooted in high performance and boasts innovation and the highest quality standards. Its' executive leadership and governing board continue to...
-
As Lush UK implemented the change, how could it measure the effects of leaving and then returning to social media on the achievement of its marketing goals? Lush Fresh Handmade Cosmetics UK sells...
-
Discuss the Extraversion aspect of the five factor model of personality.
-
After graduating from college in May 2018, Ryan Crews started his career in finance at the W&T Corporation, a small- to medium-sized warehouse distributor in Nashville, Tennessee. The company was...
-
What an example STAR (Situation, Task, Action, Result.) question for a Accounting example on how there was a time when you had a disagreement with someone. As for context use a excel project?
-
Find a trade (amount of wine exchanged for amount cheese) that makes both countries better off in both goods relative to when no trade was allowed. What is the consumption point in each country with...
-
Describe the five P's in detail (product, price, place, promotion, and people for Panera Bread.
-
Carol charged her credit card 10,000 dollars on December 31, 2011. The monthly interest rate charged by the credit card company is 1% of the unpaid balance at the end of each month. Carol wishes to...
-
Bonnie and Clyde get married and buy a home for $25,000. After 5 years, they file for divorce. As part of the divorce decree, Bonnie can live in the home until she reaches the age of 65, at which...
-
The following items were displayed in the statement of affairs for Lubbock Company: Fully secured liabilities ......... $90,000 Partially secured liabilities ....... 12,000 Unsecured liabilities...
-
The following transactions are for Pickard Construction Company: a. The firm bought equipment for $64,000 on credit. b. The firm purchased land for $450,000, $160,000 of which was paid in cash and a...
-
Bradbury Corp. reported the following asset values in 2011 and 2012: In addition, Bradbury had sales of $3,000,000 in 2012. Cost of goods sold for the year was $1,800,000. As of the end of 2011, the...
-
This coming April, you will be faced with preparing your first tax return since mom and dad said you are now on your own. As you review the IRS regulations, you notice several differences from what...
-
The responses most likely to be associated with use of a force-coercion change strategy are best described as __________. (a) internalized commitment (b) temporary compliance (c) passive cooptation...
-
The assessment center approach to employee selection relies heavily on ____________ to evaluate a candidates job skills. (a) intelligence tests (b) simulations and experiential exercises (c) 360...
-
Which of the following questions can an interviewer legally ask a job candidate during a telephone interview? (a) Are you pregnant or planning to soon start a family? (b) What skills do you have that...

Study smarter with the SolutionInn App


