Why should Ne be less soluble in water than O2? Please choose the answer that best...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
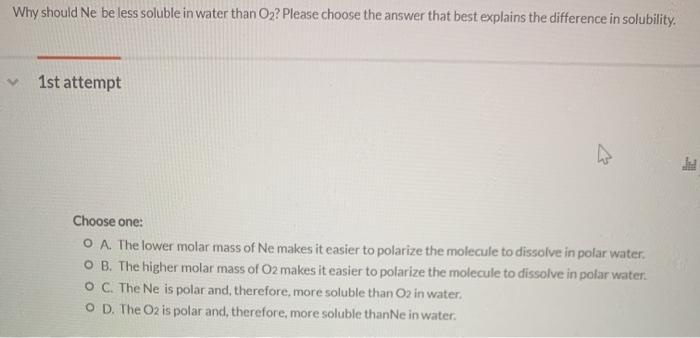
Why should Ne be less soluble in water than O2? Please choose the answer that best explains the difference in solubility. 1st attempt Choose one: O A. The lower molar mass of Ne makes it easier to polarize the molecule to dissolve in polar water. O B. The higher molar mass of O2 makes it easier to polarize the molecule to dissolve in polar water. O C. The Ne is polar and, therefore, more soluble than O2 in water. O D. The O2 is polar and, therefore, more soluble thanNe in water. Why should Ne be less soluble in water than O2? Please choose the answer that best explains the difference in solubility. 1st attempt Choose one: O A. The lower molar mass of Ne makes it easier to polarize the molecule to dissolve in polar water. O B. The higher molar mass of O2 makes it easier to polarize the molecule to dissolve in polar water. O C. The Ne is polar and, therefore, more soluble than O2 in water. O D. The O2 is polar and, therefore, more soluble thanNe in water.
Expert Answer:
Answer rating: 100% (QA)
The solubility of any covalent compound is based on the intermolecular forces of attraction More so... View the full answer

Related Book For 

Posted Date:
Students also viewed these economics questions
-
Explain how mortgage securitization makes it easier to borrow money to buy a house but harder to deal with when a house is sold for a loss?
-
Please select the answer that best applies to each question below. Each answer applies only once A thief sues Jane Homeowner after he injures himself on the broken stairs of Jane's house. The ACME...
-
For the following question, select the answer that best matches the transaction from the list below, the current year end is 12/31/2015: A. Accrual only B. Deferral only C. Accrual and Adjustment D....
-
When developing a survey instrument for a cross-country study, market researchers often need to construct a scale (e. g., a 7-point disagree/agree scale). What are the major items that one should be...
-
The responses of a sample of 1000 adults who were asked what type of corrective lenses they wore are shown in the table at the left. Type of lenses Frequency, f Contacts ............. 40 Eyeglasses...
-
Destin Products makes digital watches. Destin is preparing a product-life cycle budget for a new watch, MX3. Development on the new watch is to start shortly. Estimates for MX3 are as follows: 1....
-
Find a recent newspaper article or television report about a statistical study on a topic that you find interesting. Write a short report applying each of the eight guidelines given in this section....
-
On January 1, 2017, Shellenburger Inc. had the following stockholders equity account balances. Common Stock , no-par value (500,000 shares issued) . $1,500,000 Common Stock Dividends Distributable...
-
A primary school needs to arrange their students to sit for the National Assessment ProgramLiteracy and Numeracy test in multiple exam halls at Griffith University. Each school class has 25 students....
-
A parent company acquired 70% of the stock of a subsidiary company on January 1, 2017, for $610,000. On this date, the balances of the subsidiarys stockholders equity accounts were Common Stock,...
-
A and B share profits and losses equally. They admit C as an equal partner and goodwill was valued as Rs.30,000. C is to bring in 30,000 as his capital and necessary cash towards his share of...
-
Explain what arguments the plaintiff (Mr. Henderson) could raise to the court to substantiate his lawsuit against the Condo Corporations. 2. Explain what arguments the defendant (Jefferson Property...
-
What is the definition of planning? What is the meaning of strategic planning? What is the intent of operational planning? Explain and contrast how they differ. 3-4 paragraphs. references
-
What are the fundamental distinctions between prokaryotic organisms and their eukaryotic counterparts, particularly concerning cellular structure and organizational complexity?
-
- What other indicators of recession do you have to share? Did sales of the goods and services produced and provided by your employers increase or decrease during the last recession?
-
Should we try and separate conduct risk and compliance risk? And why? And How?
-
As a method of citation , , what is AMA represent?
-
Explain briefly what is meant by electronic data interchange (EDI). How does EDI affect a companys audit trail?
-
When compared to people of your grandparents generation, you can expect the net present value of Social Security to be a. Much better. b. About the same. c. Slightly worse. d. Much worse.
-
The FY 2016 federal budget was around a. $4 million. b. $4 billion. c. $4 trillion. d. $4 quadrillion.
-
Which one of the following communities would likely see the greatest economic impact from a new casino? a. Plain-field, IN (just outside Indianapolis) b. Gary, IN (outside Chicago and already has...
-
Last year, Miley decided to terminate the S corporation election of her solely owned corporation on October 17, 2018 (effective immediately), in preparation for taking it public. At the time of the...
-
You have been assigned to compute the income tax provision for Motown Memories Inc. (MM) as of December 31, 2019. The companys income statement for 2019 is provided below: MM prepared the following...
-
Chandra was the sole shareholder of Pet Emporium, which was originally formed as an S corporation. When Pet Emporium terminated its S election on August 31, 2018, Chandra had a stock basis and an...

Study smarter with the SolutionInn App


